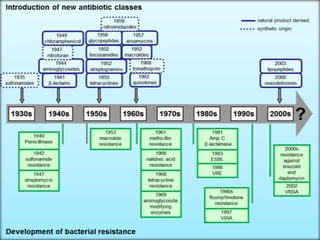
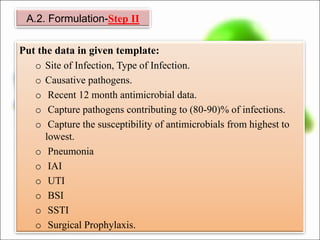
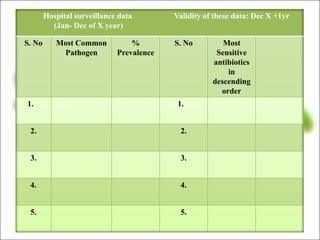
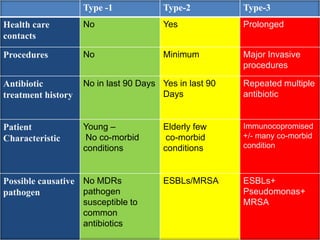
This document outlines recommendations for developing and implementing an antibiotic policy in hospitals. It discusses formulating the policy based on local antimicrobial susceptibility data. The policy should include guidelines for empiric, prophylactic, and definitive antibiotic therapy along with restricted and reserve drug lists. It recommends establishing an antibiotic management team to monitor implementation and assess outcomes, such as antibiotic consumption. Regular review and updating of the policy based on new clinical and susceptibility data is also advised. The overall goal is to optimize antibiotic use and slow the development of antimicrobial resistance.