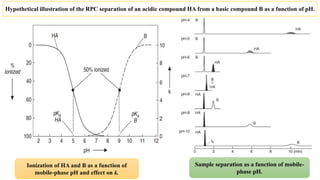
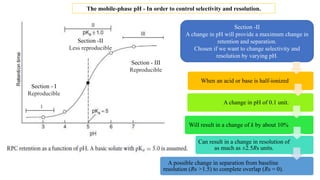
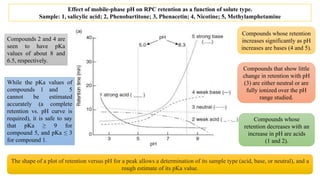
The document discusses the role of acid-base equilibria and pKa values in reversed-phase chromatography (RPC) method development. It explains how changes in mobile-phase pH can significantly affect the retention and separation of compounds, particularly when dealing with acids and bases. Additionally, it emphasizes the need to carefully control pH to achieve desired selectivity and resolution in sample analysis.

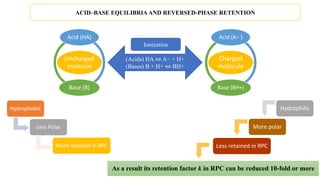
![Acids
HA ⇔ A− + H+
Bases
B + H+ ⇔ BH+
When the mobile-phase
pH is increased
When mobile-phase pH
decreases
Bases gain a proton
and become ionized
Acids lose a proton and
become ionized
Acid Bases
(Acids) Ka = [A−][H+]
[HA]
(Bases) Ka = [B][H+]
[BH+]
Acidity constant Ka
𝑨𝑪𝑰𝑫 𝑷 𝑲𝒂 = 𝒑𝑯 − 𝐥𝐨𝐠
𝑨−
𝑯𝑨
𝑩𝑨𝑺𝑬𝑺 𝑷 𝑲𝒂 = 𝒑𝑯 − 𝐥𝐨𝐠
𝑩
𝑩𝑯 +
Henderson–Hasselbalch equation
pKa value = −logKa
ACID–BASE EQUILIBRIA AND REVERSED-PHASE RETENTION](https://image.slidesharecdn.com/acidbaseequilibriaandreversedphaseretention-190901175811/85/Acid-base-equilibria-and-reversed-phase-retention-3-320.jpg)