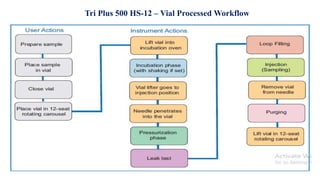
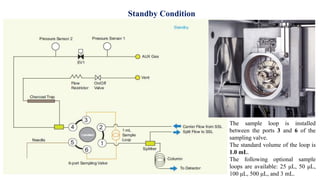
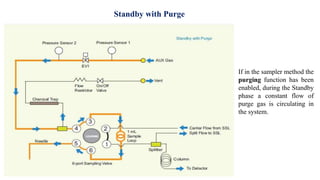
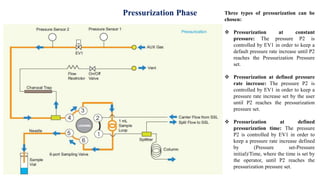
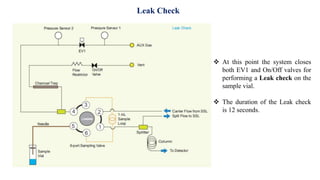
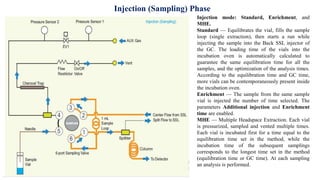
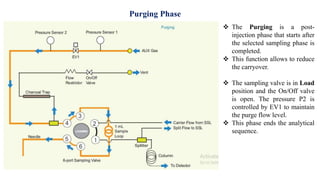
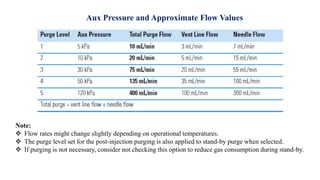
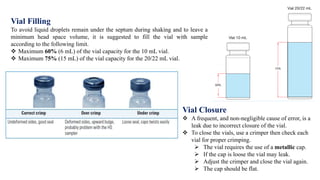
This document provides information on the working of the TriPlus 500 HS automated headspace sampler. It describes the different phases of the sampler's workflow including pressurization, injection, sampling, and purging. It also discusses parameters that can be set for each phase as well as considerations for temperature, carrier gas flow, auxiliary gas pressure, sample vials, shaking conditions, and more. The goal is to optimize the headspace sampler's settings and parameters to achieve the desired analytical sensitivity.