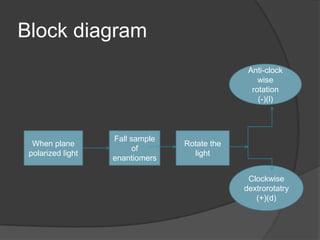
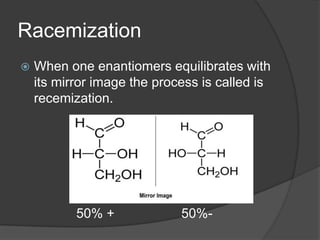
This document discusses chirality and enantiomers. It explains that chiral compounds contain a stereocenter, which is an atom connected to four different substituents, and thus exist as two non-superimposable mirror images called enantiomers. The document outlines methods for distinguishing between enantiomers using R/S notation at stereocenters, their different optical rotations when interacting with polarized light, and various diagrammatic representations including Newman projections and Fisher projections. It also discusses racemic mixtures, racemization, and resolution as a process to separate enantiomers from a racemic mixture.