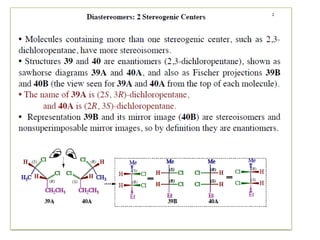
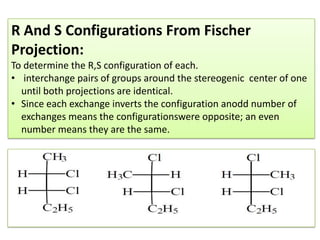
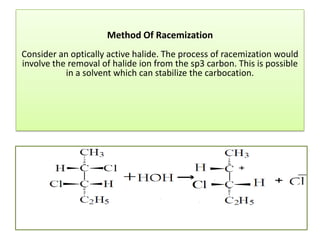
2,3-dichloropentane exhibits stereoisomerism with chiral centers that produce optical activity. It can have enantiomers and diastereomers depending on the spatial arrangement of its atoms. Racemization is the process where an optically active compound is converted to a 50/50 mixture of both enantiomers. This can be achieved by removing a halide ion from the chiral carbon to form a carbocation intermediate. Resolution separates the enantiomers from a racemic mixture using various methods like mechanical separation, biological processes, chemical reactions, or chromatography.