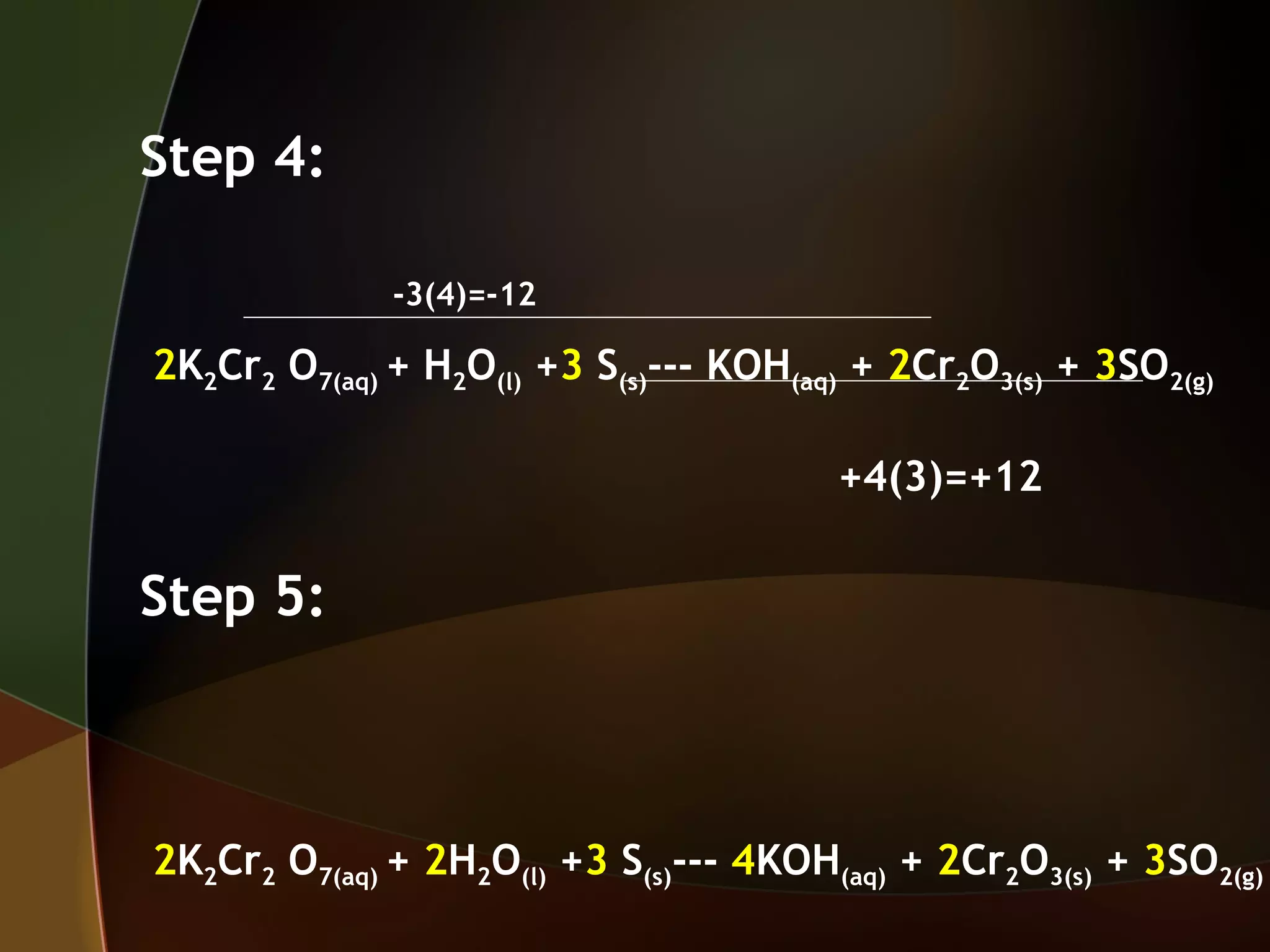
The document discusses two methods for balancing redox reactions:
1) Using oxidation number changes, which involves assigning oxidation numbers, identifying oxidized and reduced elements, and using coefficients to equalize oxidation number changes.
2) Using half-reactions, which involves writing separate half-reactions for oxidation and reduction, balancing atoms, adding electrons, and combining half-reactions to give the overall balanced equation. The half-reaction method is best for reactions in acidic or alkaline solutions.