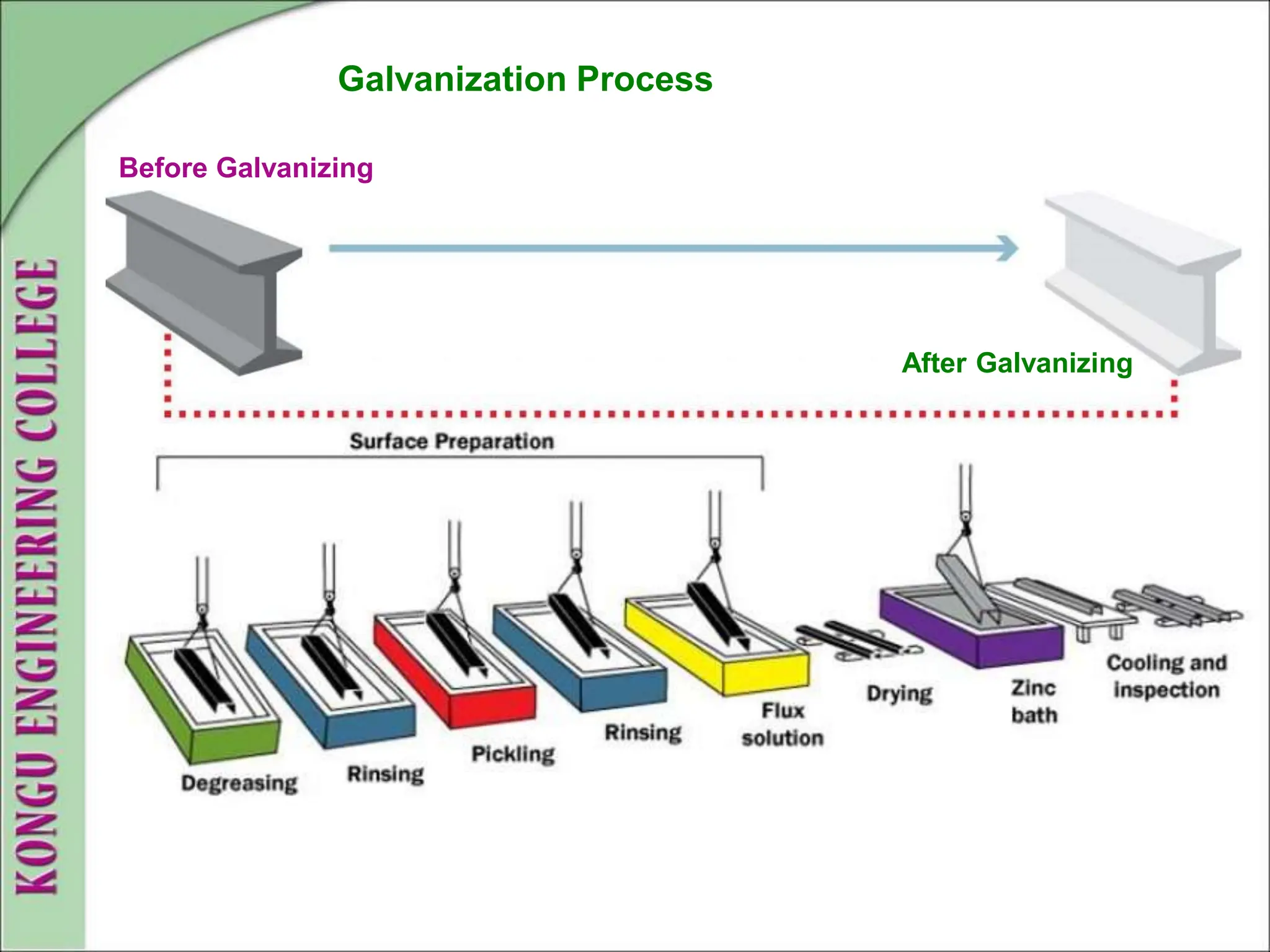
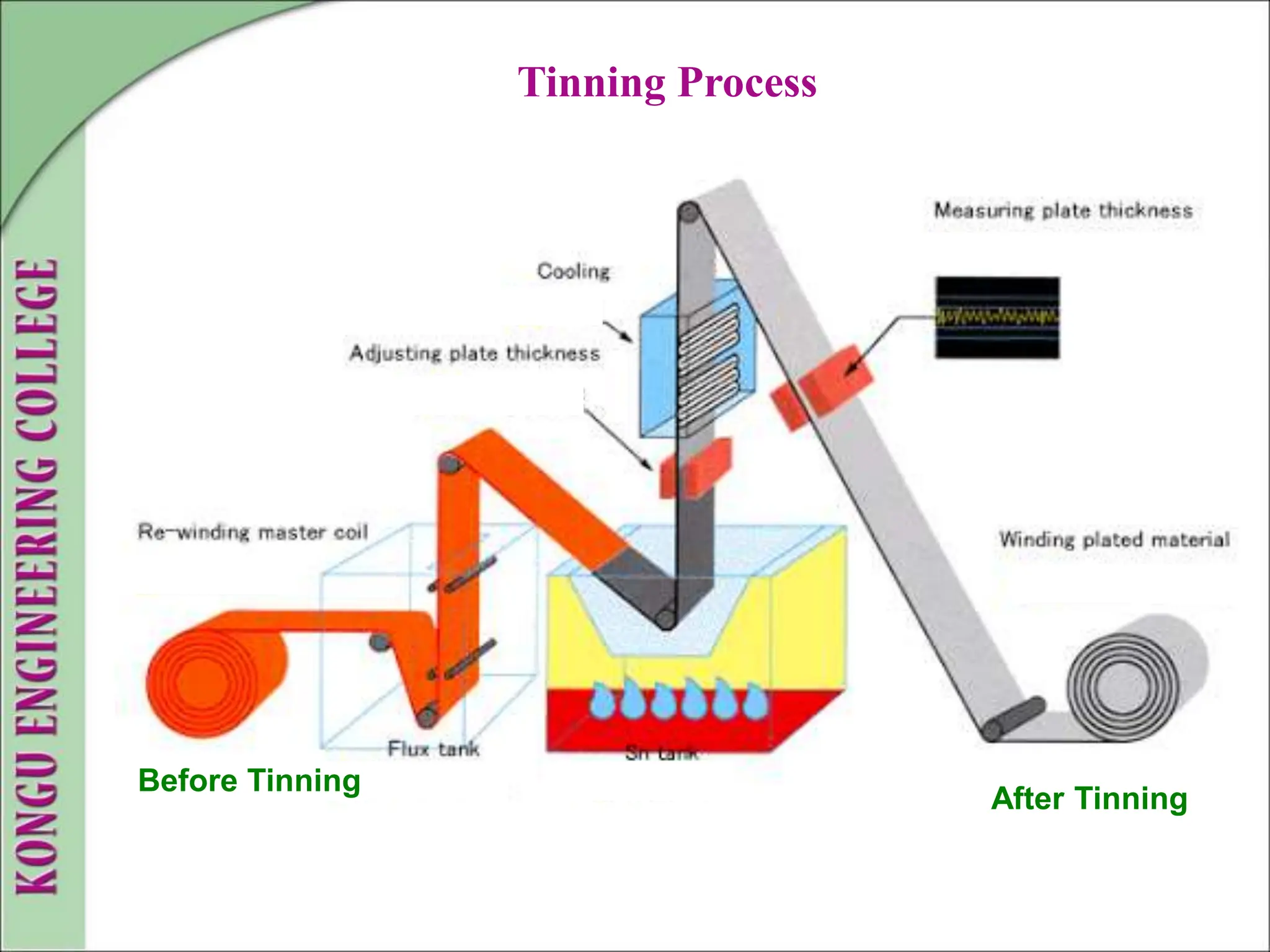
The document discusses corrosion, defining it as a destructive chemical and electrochemical reaction that degrades metals through interactions with their environment. It covers types of corrosion, factors influencing the rate of corrosion, and various control methods such as sacrificial anodic protection, corrosion inhibitors, and protective coatings. Understanding and managing corrosion is crucial due to its widespread consequences, including structural failures and economic losses.









![Types of dry corrosion
II. Corrosion by Other Gases
Other gases such as Cl2,SO2,H2S,F2,CO2 & NOX. In dry atmosphere
these gases react with metal and form corrosion products.
2AgCl + Cl2 AgCl
(Non-Porous layer)
Sn + 2Cl2 SnCl4
(Volatile Layer)
Hydrogen Embrittlement (or) Hydrogen Corrosion
At high temperature
Fe + H2S FeS + 2H
[H] + [H] H2
FeS is a Porous Layer and corrosion is a continuous process.
Atomic hydrogen penetrates and occupies the voids of the metal. Then
develops pressure which leads to cracking of the metal.
This is known as Hydrogen Embrittlement (or) Hydrogen Corrosion](https://image.slidesharecdn.com/22cyt12chemistryforcomputersystemsunit-ii-corrosionitscontrolmethods-240408042811-88ce9172/75/22CYT12-Chemistry-for-Computer-Systems_Unit-II-Corrosion-its-Control-Methods-ppt-10-2048.jpg)