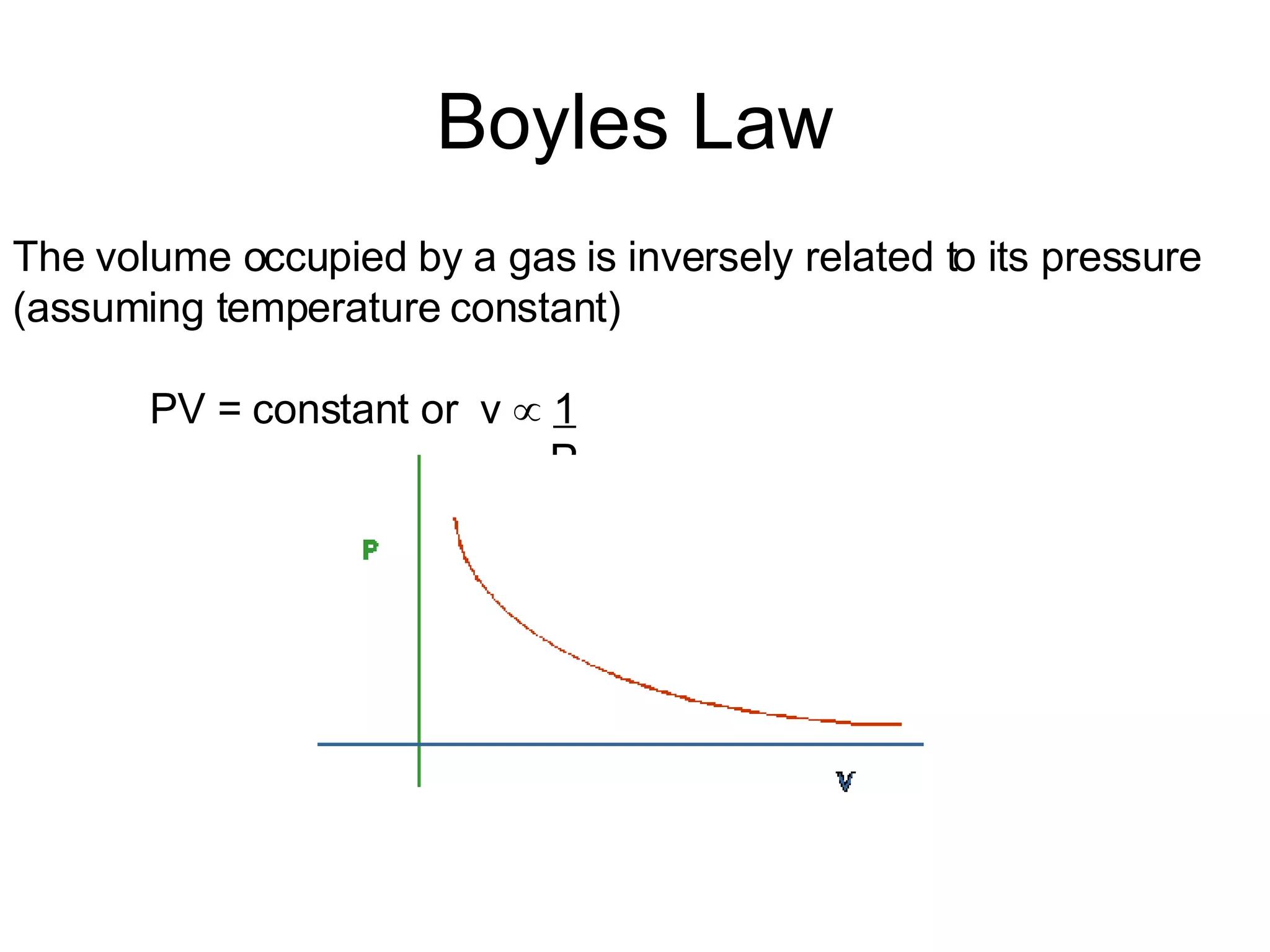
The document summarizes 12 gas laws including Boyle's law, Charles' law, Avogadro's law, and the ideal gas law. It provides examples of calculations using these laws to determine moles of gas, volumes at different temperatures and pressures, and identities of gases based on density. Key formulas covered are PV=nRT, relationships between volume, pressure and temperature, and stoichiometric calculations using gas volumes.