Bacterial Plasmids.pptx
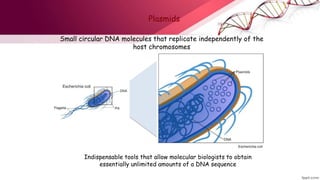
- 1. Plasmids Indispensable tools that allow molecular biologists to obtain essentially unlimited amounts of a DNA sequence Small circular DNA molecules that replicate independently of the host chromosomes
- 2. Goals • Learn about Plasmids, • types, and • mode of their replication.
- 3. Terminology • Curing: process of eliminating plasmid from bacteria • Episome: integration of plasmid with the chromosome
- 4. Bacterial Plasmids in Nature 1. Occur naturally in bacteria and usually carry genes that are useful but not essential to survival: e.g. genes which make bacteria resistant to antibiotics. 2. Plasmids are released by dead bacteria and absorbed by those still living thus genetic information is exchanged (sexual reproduction?).
- 5. Bacterial Plasmids in Nature 3. Some plasmids even contain genes that build a transfer tube between bacteria. 4. There can be as many as several hundred copies of a single plasmid in each bacteria.
- 6. Plasmid Structure 1. Plasmids only need an “origin or replication” and a “useful” gene to be considered complete. 2. Molecular biologists have been able to “insert” custom built restriction sites into many plasmids so they can be used to “insert” DNA fragments from other genes into them and thus have a way to propagate those DNA pieces.
- 7. Classification On the basis of ability to perform conjugation: Conjugative/self transmissible plasmid Non conjugative plasmid Based on compatibility b/w plasmid: Compatible Incompatible Based on function: Fertility/F plasmid: contain tra gene: sex pili expression Resistance/R plasmid Col plasmid Virulence plasmid Metabolic plasmid
- 8. • Usual double stranded closed cirle plasmids • SS stranded circular DNA plasmids- Streptomyces, Clostridium • Linear double stranded DNA plasmids in several bacteria and eukaryotic sources-Borrelia hermstii • RNA plasimd – viroids are specialized in ss circular RNA plasmids carrying no genes • Bipartite, linear ds RNA elements in yeast called killer factors Based on structure
- 9. Classification • Plasmids may be classified in a number of ways. • Plasmids can be broadly classified into conjugative plasmids and non-conjugative plasmids. Conjugative plasmids • Conjugative plasmids contain a set of transfer or tra genes which promote sexual conjugation between different cells. • In the complex process of conjugation, plasmid may be transferred from one bacterium to another via sex pili encoded by some of the tra genes. Non-conjugative plasmids • Non-conjugative plasmids are incapable of initiating conjugation, hence they can be transferred only with the assistance of conjugative plasmids. • An intermediate class of plasmids are mobilizable, and carry only a subset of the genes required for transfer. • They can parasitize a conjugative plasmid, transferring at high frequency only in its presence.
- 10. Incompatibility groups • Plasmids can also be classified into incompatibility groups. • A microbe can harbor different types of plasmids, however, different plasmids can only exist in a single bacterial cell if they are compatible. • If two plasmids are not compatible, one or the other will be rapidly lost from the cell. • Different plasmids may therefore be assigned to different incompatibility groups depending on whether they can coexist together. • Incompatible plasmids normally share the same replication or partition mechanisms and can thus not be kept together in a single cell. • Plasmids that are able to coexist in the same cell do not interfere with each other’s replication
- 11. Plasmid Biology 1. Plasmid structure 2. Plasmid replication and copy number control 3. Plasmid transfer 4. Plasmids as tools 5. F plasmids
- 12. 1. Extrachromosomal DNA, usually circular-parasite? 2. Usually encode ancillary functions for in vitro growth 3. Can be essential for specific environments: virulence, antibiotics resistance, use of unusual nutrients, production of bacteriocins (colicins) 4. Must be a replicon - self-replicating genetic unit 5. Plasmid DNA must replicate every time host cell divides or it will be lost a. DNA replication b. partitioning (making sure each progeny cells receives a plasmid) 6. High copy plasmids are usually small; low copy plasmids can be large 7. Partitioning is strictly controlled for low copy, but loose for high copy 8. Plasmid replication requires host cell functions 9. Copy number is regulated by initiation of plasmid replication 10. Plasmids are incompatible when they cannot be stably maintained in the same cell because they interfere with each other’s replication. Functions
- 13. Also, virulence plasmids from Salmonella, Shigella, Yersinia, B. anthracis, E.coli, and others.
- 14. Based on phenotypes Enterotoxin production - Enterobacteria Ent P 307 Heavy metal resistance - Pseudomonas sps FP2 Tumor induction - Agrobacterium tumefaciens U.V. protection - plasmid R46 Senescence - Neurospora crassa Bacteriocin production , HS production , degradation of aromatic compounds Phage Resistance Sugar fermentations
- 16. Plasmid replication 1. Plasmid replication requires host DNA replication machinery. 2. Most wild plasmids carry genes needed for transfer and copy number control. 3. All self replication plasmids have a oriV: origin of replication 4. Some plasmids carry and oriT: origin of transfer. These plasmids will also carry functions needed to be mobilized or mob genes. 5. Plasmid segregation is maintained by a par locus-a partition locus that ensures each daughter cells gets on plasmid. Not all plasmids have such sequences. 6. There are 5 main “incompatibility” groups of plasmid replication. Not all plasmids can live with each other. 7. Agents that disrupt DNA replication destabilize or cure plasmids from cells.
- 17. Types of plasmid replications • Well-known replication systems of circular plasmids include a) Theta-type replication, b) Rolling-circle replication, and c) Strand displacement-type replication
- 18. Theta type replication • Theta mode of replication is similar to chromosomal replication. • There is the synthesis of leading- and lagging-strand. • Lagging-strand is discontinuous. • No DNA breaks are required for this mode of replication. • There is the formation of bubbles in the early stages of replication. • It resembles the Greek letter θ. • Theta replication is of 4 types: – θ class A – θ class B – θ class C – θ class D
- 19. Rolling circle model • Rolling circle replication mechanism is specific to bacteriophage family M13 and the fertility F factor which encodes for sex pili formation during recombination by means of conjugation. • Fragments smaller than 10 kilo base (Kb) usually replicate by this replication mechanism as reported in some gram positive bacteria. • It allows the transfer of single stranded replication product at a faster rate to the recipient cell through pilus as in case of fertility factor or to the membrane in case of phage • Rolling circle occurs to a covalently closed circular piece of double-stranded DNA. • A nick is produced in one of the strands by enzyme nickases creating a 5’ phosphate and a 3’ hydroxyl. • Free 3’ hydroxyl will be used by DNA polymerase to make new DNA pushing the old nicked strand off of the template DNA
- 20. Single strand displacement model • Replication begins at one end of linear genome (= chromosome), eg: in adenovirus • Only one of the two strands i.e. the strand oriented in 3’ to 5’ direction of the DNA duplex is replicated in a continuous manner. • This produces a DNA duplex and a single strand (the 5’ to 3’ strand). • The single strand base pairs at its ends to form a circle: replication of this strand now proceeds in a continuous manner • In adenovirus, a protein attached to the 5’ end provides the primer function • This protein has a cytosine residue attached to it: the free 3’-OH of this C is used as primer.
- 21. Site-directed mutation: Suicide plasmds 1. Plasmid must be unable to replicate without essential replication proteins provide in trans. 2. It helps if the plasmid can be mobilized-oriT required 3. Need a selectable marker 4. Large or small region of homologous DNA cloned that will integrate into the chromosomal target. 5. Need a counter selection method to kill the donor cells 6. Screen for what you think is correct. Also, merodiploid reporter strains can be constructed in this manner 1. Make a lacZ fusion to your promoter of interest 2. Clone into a suicide plasmid 3. Mate into recipient. 4. Resulting strain will harbor a duplication of the promoter region:lacZ and still have a functional copy of the gene. Why would this be important? Plasmids as genetic tools: Construction of Mutants
- 22. Plasmids can be manipulated easily in the laboratory 1. Plasmids can be collected from bacteria. 2. Restriction enzymes can be purified and used to identify and cut out specific sequences of DNA along with the plasmid vector. 3. Ligase (enzyme) can be purified and used to “glue” pieces of DNA together. 4. Bacteria can be transformed by taking in plasmids given to them.
- 23. Transforming Bacteria • When a bacterial cell takes in a plasmid from the environment, it has new DNA (and therefore, new traits). • Scientists say that the bacteria has been transformed.
- 24. How to produce human insulin from bacteria and become a multimillionaire 1. Isolate plasmid DNA from bacteria and insulin gene from human. 2. Cut both DNAs with the same restriction enzyme. 3. Mix the DNA together with ligase. 4. Insert the new DNA into bacteria (transformation). 5. Use antibiotics to kill any bacteria without the plasmid. 6. Grow bacteria and harvest the insulin. Insulin Purification Insulin gene Human DNA
- 25. Transfer of plasmids can occur by four routes 1. Cell fusion 2. Transformation 3. Transduction 4. Conjugation
- 27. Hfr X F- crosses
- 28. This mechanism explains the characteristics of F' X F- crosses. The F- becomes F', the F' remains F' and the is high frequency transfer of donor genes on the F' but low frequency transfer of other donor chromosomal genes.
- 29. 1. large (100 kb) 2. low copy (1-2 copies/cell) 3. self transmissible 4. requires protein synthesis (chloramphenicol-sensitive) 5. repE gene encodes RepE protein 6. RepE protein binds to origin of replication (oriS) and initiates DNA replication 7. RepE binds to the repE promoter and activates transcription 8. RepE binds to the copA/incC locus binding copies of F together via RepE – inhibiting replication (coupling) F-plasmid
- 30. F-transfer at fine detail
- 31. How are plasmids constructed? What functional elements are found in our yeast overexpression plasmids? How are plasmids purified?
- 32. Plasmids used in molecular biology have been constructed in the lab Molecular cloning Enzymes are used to insert desired pieces of foreign DNA into plasmids Bacterial cells are transformed with the plasmids. Copies of the plasmids are purified from bacteria.
- 33. Shuttle vectors have origins of replication and selectable markers for propagation in both bacteria and yeast We are using plasmids that have been termed shuttle vectors, because they can be propagated in either bacteria or yeast Plasmids are propagated in bacteria, which grow quickly and maintain multiple copies of the plasmids non-pathogenic strain of Escherichia coli Saccharomyces cerevisiae deletion strains Plasmid-encoded genes are expressed in yeast, and phenotypes are analyzed
- 34. How are plasmids constructed? What functional elements are found in our yeast overexpression plasmids? How are plasmids purified?
- 35. How are plasmids constructed? What functional elements are found in our yeast overexpression plasmids? How are plasmids purified?
- 36. Plasmids are much smaller than bacterial chromosomes Plasmids are supercoiled in their native form Supercoiling allows plasmids to renature quickly after they are denatured Plasmid purification is based on their distinctive physical properties Plasmids used in molecular biology are highly engineered and contain elements of use to researchers
- 37. Plasmid purification from bacteria relies on their unique physical properties Bacterial cell with plasmids contains MANY different, well-folded proteins 1-2 copies of large (>Mbp) , circular bacterial DNA complexed with proteins Multiple copies of small (5-15 kbp) plasmids Purification involves sequential denaturation and renaturation steps
- 38. Cells are first treated with base and a detergent breaks open membrane and denatures both DNA and proteins Proteins denature irreversibly Chromosomal DNA denatures—will have difficulty renaturing because of its length and many proteins complexed to it Plasmids denature, but strands stay together because of supercoiling
- 39. Extract is neutralized to allow DNA molecules to renature Plasmids renature and are suspended in the SUPERNATANT following centrifugation Proteins and chromosomal DNA form aggregate irreversibly, forming a PRECIPITATE that can be collected by centrifugation When purifying plasmids, use a micropipette to remove the supernatant for further processing steps
- 40. Isolation and purification of Plasmid MiniPrep procedure
- 41. PLASMID ISOLATION AND ANALYSIS: Part II Pellet 2 ml of overnight cell suspension by adding 1 ml (20 drops) of overnight culture suspension into a 1.5 ml reaction tube. Spin for 1 minute at 10,000 rpm in a microcentrifuge (MicroV). Plasmid Purification and Isolation Step 1
- 42. PLASMID ISOLATION AND ANALYSIS: Part II A pellet should form in the bottom of the tube. Drain off the liquid into a designated container and gently tap the tube on a paper towel to remove the excess broth. Repeat the procedure with a second 1 ml volume of overnight culture suspension. Plasmid Purification and Isolation Step 1 (cont.)
- 43. PLASMID ISOLATION AND ANALYSIS: Part II Use a small needle pipette to add the contents of the tube marked “Cell Lysis Solution”(P2) to the Resuspension tube and mix by inverting the tube 4 times. The cell suspension should appear clear. Plasmid Purification and Isolation Step 3
- 44. PLASMID ISOLATION AND ANALYSIS: Part II Use a small needle pipette to add the contents of the tube marked “Neutralization Solution” (N3) to the Resuspension tube and mix by inverting the tube 4 times. Plasmid Purification and Isolation Step 4
- 45. PLASMID ISOLATION AND ANALYSIS: Part II Centrifuge the tube containing the lysate at 10,000 rpm for 10 minutes. Plasmid Purification and Isolation Step 5
- 46. PLASMID ISOLATION AND ANALYSIS: Part II While the lysate is in the centrifuge, label two 1.5 ml tubes. Label the lid and side of one tube “L”. Plasmid Purification and Isolation Step 6
- 47. PLASMID ISOLATION AND ANALYSIS: Part II Label the lid and side of the second tube “M.” Remove the lid from the tube with a scissors. Place the lid back on the tube and hold until Part III, step 7. Plasmid Purification and Isolation Step 7
- 48. PLASMID ISOLATION AND ANALYSIS: Part II Use the small needle pipette to carefully remove all of the clear lysate/supernate from the miniprep in step 5. Do not remove any of the precipitate. Wipe the end of the pipette with a chem wipe or kleenex to remove any protein/chromosomal DNA precipitate. Plasmid Purification and Isolation Step 8
- 49. PLASMID ISOLATION AND ANALYSIS: Part II Pipette the clear lysate/supernate into the tube marked “L.” At this point you can choose to store the tube in the refrigerator until the next day or, if time permits, continue to Part III. Plasmid Purification and Isolation Step 9
- 50. Zyppy purification kit use multiple steps to purify plasmids Alkaline lysis Neutralization Purification of plasmid DNA on a silica resin Elution of purified DNA from he silica resin Let's look at the individual steps……………..
- 51. CsCl gradient with ethidium bromide and UV light. Three forms of plasmid DNA “Old School method of purifying plasmid”
- 52. 1 Transformed E. coli cultures are concentrated by centrifugation 2. The cell pellet is resuspended in 600 µL TE buffer by vortexing 3. 100 µL of 7X Blue Zyppy lysis buffer is added 0.1 N NaOH in buffer lyses the cells GENTLY mix the contents by inverting the tube 4-6 times Solution changes from cloudy to clear when cells are lysed Warning: too much mechanical agitation can shear chromosomal DNA Alkaline lysis
- 53. Neutralization 4. Add 350 µL yellow Zyppy Neutralization buffer Mix by inverting several times A heavy precipitate will begin to form immediately! The initial “glop” will become more granular when neutralization is complete—but don’t overdo it! The precipitate contains denatured proteins and the denatured chromosomal DNA. 5. Spin down the denatured molecules for 3 minutes at top speed. CAREFULLY remove the supernatant containing the plasmid – Don't be greedy! Purity is preferred to yield!
- 54. Purification on Zyppy silica resin 6. Apply the supernatant to the spin column. Place the column in the collection tube. Centrifuge the column for ~15 seconds at top speed. 7. Discard the flow through in the collection tube. Add 200 µL Zyppy EndoWash. Centrifuge ~15 sec. EndoWash contains guanidine hydrochloride and isopropanol. It removes contaminating proteins that are bound to the resin. 8. Discard the flow through in the collection tube. Add 400 µL Column Wash. Centrifuge 1 min.
- 55. Plasmid Elution 9. Transfer the column to a clean, LABELED microcentrifuge tube 10. Add 50 µL TE buffer directly on top of the column. Allow the column to stand upright in the test tube for ~10 min. (Plasmid is being eluted.) 11. Spin the column for 30 seconds. Plasmid DNA will be collected in the microcentrifuge tube. Pure plasmid DNA collects here!
