Std xi-chem-ch1-concepts-units-of-measurement
•Download as PPTX, PDF•
1 like•470 views
Units of measurement in Chemistry
Report
Share
Report
Share
More Related Content
What's hot
What's hot (20)
Units of measurements( standard system for weight, length and volume)

Units of measurements( standard system for weight, length and volume)
Viewers also liked
Viewers also liked (20)
Std XI-Chem-Ch1-Concepts-Chemical-reactions-Stoichiometry

Std XI-Chem-Ch1-Concepts-Chemical-reactions-Stoichiometry
Std XI-Chem-Ch1-Concepts-Isotopes-Atomic-mass-Molecular-mass

Std XI-Chem-Ch1-Concepts-Isotopes-Atomic-mass-Molecular-mass
Similar to Std xi-chem-ch1-concepts-units-of-measurement
Similar to Std xi-chem-ch1-concepts-units-of-measurement (20)
Lecture Notes: EEEC6430312 Measurements And Instrumentation - Fundamentals O...

Lecture Notes: EEEC6430312 Measurements And Instrumentation - Fundamentals O...
Diploma sem 2 applied science physics-unit 1-chap 1 measurements

Diploma sem 2 applied science physics-unit 1-chap 1 measurements
Mechanics Chapter 1 units, base & derived quantities

Mechanics Chapter 1 units, base & derived quantities
More from Gurudatta Wagh
More from Gurudatta Wagh (19)
Recently uploaded
Model Call Girl in Bikash Puri Delhi reach out to us at 🔝9953056974🔝

Model Call Girl in Bikash Puri Delhi reach out to us at 🔝9953056974🔝9953056974 Low Rate Call Girls In Saket, Delhi NCR
Organic Name Reactions for the students and aspirants of Chemistry12th.pptx

Organic Name Reactions for the students and aspirants of Chemistry12th.pptxVS Mahajan Coaching Centre
call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️

call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️9953056974 Low Rate Call Girls In Saket, Delhi NCR
Recently uploaded (20)
Enzyme, Pharmaceutical Aids, Miscellaneous Last Part of Chapter no 5th.pdf

Enzyme, Pharmaceutical Aids, Miscellaneous Last Part of Chapter no 5th.pdf
ENGLISH5 QUARTER4 MODULE1 WEEK1-3 How Visual and Multimedia Elements.pptx

ENGLISH5 QUARTER4 MODULE1 WEEK1-3 How Visual and Multimedia Elements.pptx
Model Call Girl in Bikash Puri Delhi reach out to us at 🔝9953056974🔝

Model Call Girl in Bikash Puri Delhi reach out to us at 🔝9953056974🔝
History Class XII Ch. 3 Kinship, Caste and Class (1).pptx

History Class XII Ch. 3 Kinship, Caste and Class (1).pptx
Organic Name Reactions for the students and aspirants of Chemistry12th.pptx

Organic Name Reactions for the students and aspirants of Chemistry12th.pptx
call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️

call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️
Call Girls in Dwarka Mor Delhi Contact Us 9654467111

Call Girls in Dwarka Mor Delhi Contact Us 9654467111
Introduction to ArtificiaI Intelligence in Higher Education

Introduction to ArtificiaI Intelligence in Higher Education
Std xi-chem-ch1-concepts-units-of-measurement
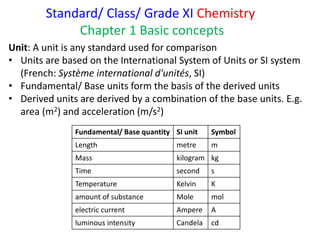
- 1. Standard/ Class/ Grade XI Chemistry Chapter 1 Basic concepts Fundamental/ Base quantity SI unit Symbol Length metre m Mass kilogram kg Time second s Temperature Kelvin K amount of substance Mole mol electric current Ampere A luminous intensity Candela cd Unit: A unit is any standard used for comparison • Units are based on the International System of Units or SI system (French: Système international d'unités, SI) • Fundamental/ Base units form the basis of the derived units • Derived units are derived by a combination of the base units. E.g. area (m2) and acceleration (m/s2)
- 2. The measurements given below range from small to big. They are arranged in an ascending order. Femtometre on the extreme left is smaller than (<) petametre on the extreme right. femtometre fm (10-15 m) < picometre pm (10-12 m) < nanometre nm (10-9 m) < micrometre µm (10-6 m) < millimetre mm (10-3 m) < centimetre cm (10-2 m) < decimetre dm (10-1 m) < metre m (100 m) < decametre dam (101 m) < hectometre hm (102 m) < kilometre km (103 m) < megametre Mm (106 m) < gigametre Gm (109 m) < terametre Tm (1012 m) < petametre Pm (1015 m) Sizes: atom and subatomic particles
