Ionic bonding
•Download as PPTX, PDF•
0 likes•393 views
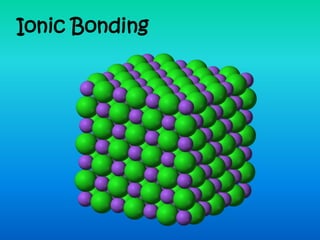
Ionic bonding occurs when atoms combine to form ions that are held together by electrostatic attraction between oppositely charged ions. Metals bond ionically with nonmetals by transferring electrons from the metal to the nonmetal, forming cations and anions that bond to create an electrically neutral compound. Examples provided are sodium (Na+) bonding with fluorine (F-), magnesium (Mg2+) bonding with chlorine (Cl-), and aluminum (Al3+) bonding with oxygen (O2-).
Report
Share
Report
Share
Recommended
Recommended
More Related Content
What's hot
What's hot (20)
Unit b matter and chemical change notes(compounds & properties)

Unit b matter and chemical change notes(compounds & properties)
Grade 9 chemistry, ions and writing chemical formulae

Grade 9 chemistry, ions and writing chemical formulae
Viewers also liked
Viewers also liked (12)
Chapter 6.2 : Covalent Bonding and Molecular Compounds

Chapter 6.2 : Covalent Bonding and Molecular Compounds
Similar to Ionic bonding
Similar to Ionic bonding (20)
Properties and Formation of Ionic Compounds Powerpoint

Properties and Formation of Ionic Compounds Powerpoint
More from eliebengston
More from eliebengston (20)
Recently uploaded
https://app.box.com/s/7hlvjxjalkrik7fb082xx3jk7xd7liz3TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...

TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...Nguyen Thanh Tu Collection
Making communications land - Are they received and understood as intended? webinar
Thursday 2 May 2024
A joint webinar created by the APM Enabling Change and APM People Interest Networks, this is the third of our three part series on Making Communications Land.
presented by
Ian Cribbes, Director, IMC&T Ltd
@cribbesheet
The link to the write up page and resources of this webinar:
https://www.apm.org.uk/news/making-communications-land-are-they-received-and-understood-as-intended-webinar/
Content description:
How do we ensure that what we have communicated was received and understood as we intended and how do we course correct if it has not.Making communications land - Are they received and understood as intended? we...

Making communications land - Are they received and understood as intended? we...Association for Project Management
Recently uploaded (20)
UGC NET Paper 1 Mathematical Reasoning & Aptitude.pdf

UGC NET Paper 1 Mathematical Reasoning & Aptitude.pdf
General Principles of Intellectual Property: Concepts of Intellectual Proper...

General Principles of Intellectual Property: Concepts of Intellectual Proper...
TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...

TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...
On National Teacher Day, meet the 2024-25 Kenan Fellows

On National Teacher Day, meet the 2024-25 Kenan Fellows
Jual Obat Aborsi Hongkong ( Asli No.1 ) 085657271886 Obat Penggugur Kandungan...

Jual Obat Aborsi Hongkong ( Asli No.1 ) 085657271886 Obat Penggugur Kandungan...
Mixin Classes in Odoo 17 How to Extend Models Using Mixin Classes

Mixin Classes in Odoo 17 How to Extend Models Using Mixin Classes
Making communications land - Are they received and understood as intended? we...

Making communications land - Are they received and understood as intended? we...
Unit-V; Pricing (Pharma Marketing Management).pptx

Unit-V; Pricing (Pharma Marketing Management).pptx
Asian American Pacific Islander Month DDSD 2024.pptx

Asian American Pacific Islander Month DDSD 2024.pptx
Ionic bonding
- 2. Ion Quiz… Give the Ion Symbol that will be formed for each atom: 1. Sulfur 2. Boron 3. Nitrogen 4. Magnesium
- 3. What do these compounds have in common??? • C12H22O11 sugar • C2H5OH ethanol • C6H8O6 vitamin C
- 4. Chemical Bonding When atoms combine to form new, more stable substances.
- 5. Ionic Bonding The positive and negative ions form and attract each other, holding the structure together (it’s a strong bond). + - + - Will only happen if you have one metal and one nonmetal!
- 6. Na-Sodium F-Flourine 11+ 9+ Na+1 F-1
- 7. Atoms Ionicly Bond to become a NEUTRAL compound. Bond Sodium & Fluorine: Na+1 + - F-1 Always write the Metal FIRST Compound Formed: NaF Bond Magnesium & Chlorine: Mg+2 + - Cl-1 + - Cl-1 Compound Formed: MgCl2
- 8. Bond Aluminum & Oxygen: Al+3 + - O -2 + - + - O-2 Al+3 + - + - O-2 + - Compound Formed: Al2O3