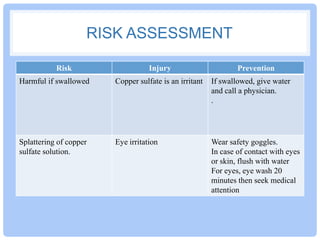
This document provides instructions for growing copper sulfate crystals through crystallization. Crystallization is a technique used to separate dissolved solids from liquids by evaporating the solvent. As the saturated solution cools, crystals will form. The size of the crystals depends on the cooling rate - fast cooling yields many small crystals while slow cooling produces fewer, larger crystals. The document outlines growing both small and large copper sulfate crystals, noting equipment needed and safety precautions to take when handling the copper sulfate solution. Drawings of the setups and pictures of the resulting crystals are to be included in an experimental report.