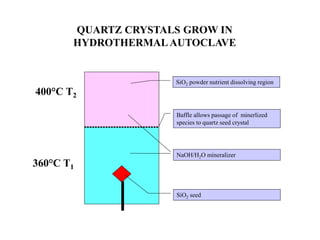
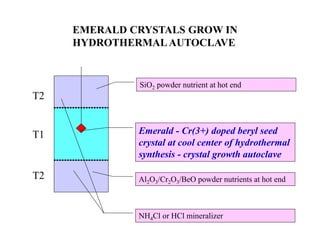
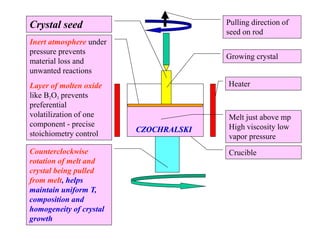
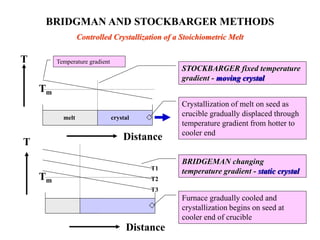
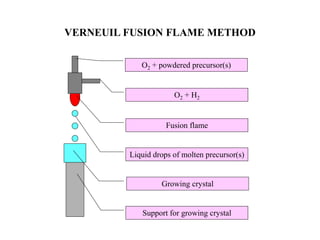
The document discusses several methods for growing single crystal materials, including Czochralski, Bridgman, Stockbarger, zone melting, and Verneuil techniques. The Czochralski method involves pulling a crystal seed from a melt of the material using precise temperature control. The Bridgman and Stockbarger methods use controlled solidification of a melt in a temperature gradient. Zone melting and hydrothermal crystal growth allow for purification or synthesis of crystals. These growth methods are important for producing large, high-quality single crystals for applications in electronics, optics, and other devices.

















![HYDROTHERMAL SYNTHESIS AND GROWTH OF
SINGLE CRYSTALS
• Ability of certain non-centrosymmetric crystals like quartz
to generate a voltage in response to applied mechanical
stress - Greek piezein - squeeze or press
• Effect reversible - piezoelectric crystals, subject to an
externally applied voltage, change shape by a small amount
• Compressive stress along [100] disturbs crystal symmetry
distorting SiO4 tetrahedra along 3-fold axis (not for [001] 2-
fold axis) creating charge asymmetry and electrical charges
across opposite crystal faces that generates a V
• Berlinite alpha-AlPO4 more polar Al-O larger than alpha-
quartz Si-O with which it is isoelectronic and isostructural -
use as a high frequency oscillator and mass monitor](https://image.slidesharecdn.com/crystalgrowth-220816085915-0a96b3f9/85/Crystal_growth-pptx-21-320.jpg)