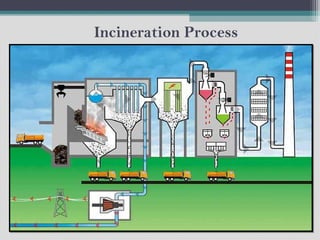
The document discusses incineration as an effective method for reducing the volume and weight of municipal solid waste (MSW) through controlled burning, achieving about 90% volume reduction and 75% weight reduction. It outlines the advantages of incineration, such as energy recovery and sterilization of waste, while also highlighting disadvantages like high capital costs and environmental concerns related to emissions. The document also briefly covers the management and treatment of biomedical and hazardous waste, including incineration, pyrolysis, and recycling.