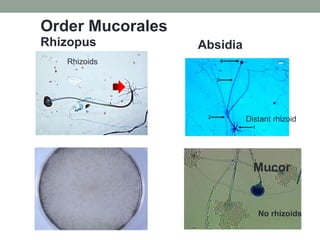
This document provides an overview of mycology (the study of fungi). It discusses different types of fungi including yeasts, molds, dimorphic fungi, and important pathogenic species. Key information covered includes fungal structure and growth characteristics, laboratory methods for fungal culture and identification, staining techniques, and histopathological findings for various fungal infections.

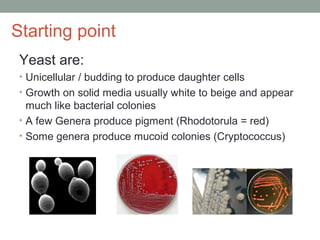
![Starting point
Molds:
• Produce filaments or hyphae
• Produce conidia [spores]
• Growth on solid media are downy, fluffy, cottony
• Most mold colonies produce pigment, which aids in
identification
hyphae
spores](https://image.slidesharecdn.com/mycology2018update-180410014152/85/Mycology-2018-Update-3-320.jpg)





![Lactophenol cotton blue [LCB] adhesive tape preparation is
used for mold identification.
LCB mounting medium consists of phenol, lactic acid,
glycerol and aniline cotton blue dye.
Clear adhesive tape touches a mold colony, picking
up fungal hyphae/conidia and pressed into one drop of LCB
on a microscope slide.
Newer (better methods)
include:
MALDI-TOF
16sRNA sequencing
Mold Identification methods](https://image.slidesharecdn.com/mycology2018update-180410014152/85/Mycology-2018-Update-9-320.jpg)

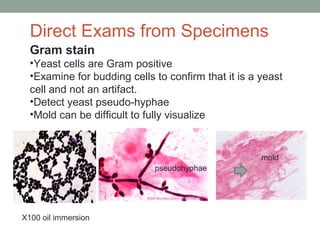



![Grocott’s Methenamine Silver Stain [GMS]
– yeast and hyphae stain grey to black. Will stain
both living and dead yeast and hyphae
• Observe the width of the hyphae, presence of hyphae
septation and angle of branching
• Observe the size and budding pattern of yeast
• Will explain on later slides how these observations can
assist in identification
Examination of fungi in fixed
tissue](https://image.slidesharecdn.com/mycology2018update-180410014152/85/Mycology-2018-Update-15-320.jpg)
![Positive staining hyphae are magenta – will also
stain structures containing
carbohydrate macromolecules (glycogen,
glycoprotein, proteoglycans) Only stains living fungi.
Periodic Acid Schiff [PAS]](https://image.slidesharecdn.com/mycology2018update-180410014152/85/Mycology-2018-Update-16-320.jpg)
![Mucicarmine stains the polysaccharide capsule of
Cryptococcus neoformans and C. gatti pink. Will
also stain mucin in fixed tissue.
Mucicarmine [Mucin] stain](https://image.slidesharecdn.com/mycology2018update-180410014152/85/Mycology-2018-Update-17-320.jpg)














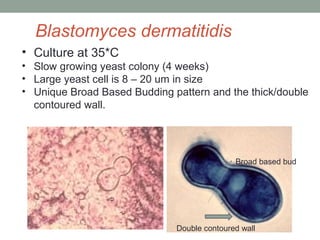






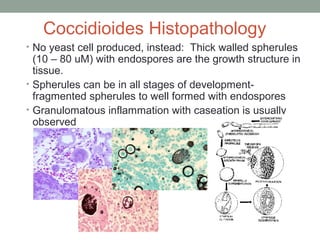



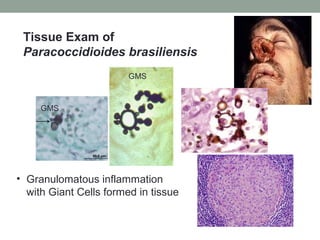







![Note: Edge of granule has thin filamentous bacteria:
How can you tell if it is Nocardia?
Nocardia is modified (partial) acid fast [PAF] positive and
is an aerobic bacteria
Actinomyces is PAF negative and grows anaerobically
Beware! Sulfur granule caused
by Actinomyces israelii looks
identical to one formed by
Nocardia.
Actinomycotic sulfur granule](https://image.slidesharecdn.com/mycology2018update-180410014152/85/Mycology-2018-Update-52-320.jpg)
![Gram stain = filamentous Gram positive bacilli
can be poorly staining and appear speckled.
Positive [red] on the Modified Kinyoun acid fast stain.
Modified (Partial) Kinyoun
acid-fast stain
Gram stain
Nocardia](https://image.slidesharecdn.com/mycology2018update-180410014152/85/Mycology-2018-Update-53-320.jpg)



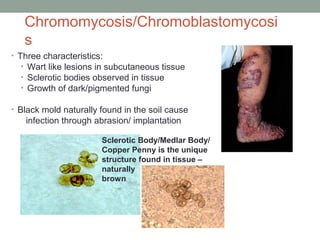

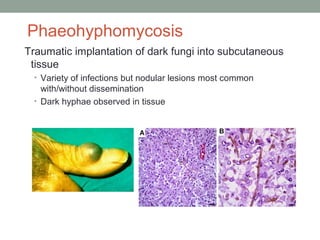
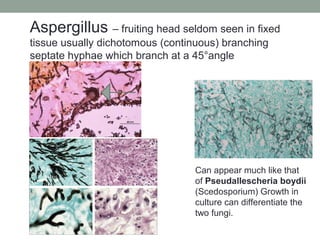
![Black mold also known as
Dematiaceous mold
-Black colored colonies and the reverse
[underside of colony] is also black
-Natural-colored brown hyphae and
spores due to melanin production
-One of the most common molds to grow
due to water damage!
Most common black molds include:
Cladophialophora carrionii
Cladophialophora bantiana
Phialophora verrucosa
Fonsecaea pedrosoi
Exophiala species
Wangiella species](https://image.slidesharecdn.com/mycology2018update-180410014152/85/Mycology-2018-Update-60-320.jpg)

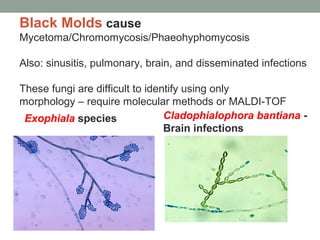



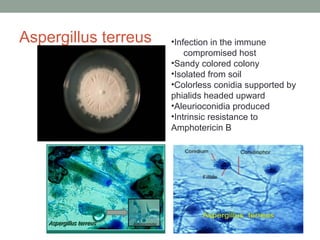
![Exserohilum rostrum
• Outbreak associated with compounded pharmaceutical
[steroid] products contaminated with dust/dirt during
manufacturing
• Injected into lumbar spine and knee joints for pain
management and led to infections:
• Meningitis
• Spinal abscess
• Synovial infections](https://image.slidesharecdn.com/mycology2018update-180410014152/85/Mycology-2018-Update-67-320.jpg)









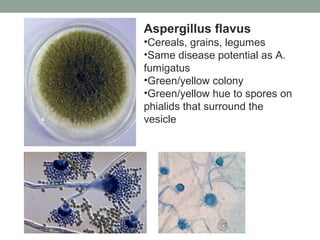
![Cryptococcus gattii – a close relative of C.
neoformans
• Isolated first from forested areas of the Pacific Northwest
(British Columbia, Washington, Oregon and California)
found in soil debris and tree species
• Infection of both normal and immune suppressed hosts
• Primarily a pulmonary disease [Cryptococcoma] but can
develop meningitis
• Culture, biochemical & staining identical to C. neoformans
• Defining reactions –
• L Canavanine glycine bromthymol blue medium –
C. gatti = blue C. neoformans = colorless
MALDI-TOF, ssequencing will also ID both species](https://image.slidesharecdn.com/mycology2018update-180410014152/85/Mycology-2018-Update-77-320.jpg)










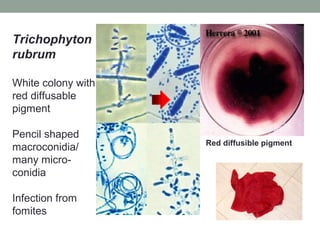
![Microsporum canis
Ringworm acquired from dog or cat
White colony/ yellow on reverse of colony
Tuberculate thick walled macroconidia [spiny
projections] Few if any microconidia](https://image.slidesharecdn.com/mycology2018update-180410014152/85/Mycology-2018-Update-88-320.jpg)