JBEI Research Highlights - February 2018
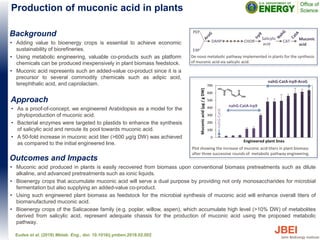
- 1. Outcomes and Impacts • Muconic acid produced in plants is easily recovered from biomass upon conventional biomass pretreatments such as dilute alkaline, and advanced pretreatments such as ionic liquids. • Bioenergy crops that accumulate muconic acid will serve a dual purpose by providing not only monosaccharides for microbial fermentation but also supplying an added-value co-product. • Using such engineered plant biomass as feedstock for the microbial synthesis of muconic acid will enhance overall titers of biomanufactured muconic acid. • Bioenergy crops of the Salicaceae family (e.g. poplar, willow, aspen), which accumulate high level (>10% DW) of metabolites derived from salicylic acid, represent adequate chassis for the production of muconic acid using the proposed metabolic pathway. Eudes et al. (2018) Metab. Eng., doi: 10.1016/j.ymben.2018.02.002 Background • Adding value to bioenergy crops is essential to achieve economic sustainability of biorefineries. • Using metabolic engineering, valuable co-products such as platform chemicals can be produced inexpensively in plant biomass feedstock. • Muconic acid represents such an added-value co-product since it is a precursor to several commodity chemicals such as adipic acid, terephthalic acid, and caprolactam. Approach • As a proof-of-concept, we engineered Arabidopsis as a model for the phytoproduction of muconic acid. • Bacterial enzymes were targeted to plastids to enhance the synthesis of salicylic acid and reroute its pool towards muconic acid. • A 50-fold increase in muconic acid titer (>600 µg/g DW) was achieved as compared to the initial engineered line. Production of muconic acid in plants 0 100 200 300 400 500 600 700 Muconicacid(µg/gDW) nahG-CatA nahG-CatA-Irp9 nahG-CatA-Irp9-AroG Plot showing the increase of muconic acid titers in plant biomass after three successive rounds of metabolic pathway engineering. PEP E4P DAHP CHOR Salicylic acid CAT Muconic acid De-novo metabolic pathway implemented in plants for the synthesis of muconic acid via salicylic acid. Engineered plant lines
- 2. Improving methyl ketone production in E. coli by heterologous expression of NADH-dependent FabG Outcomes and Impacts • The best methyl ketone performance occurred in strains expressing FabG derived from Acholeplasma laidlawii (Al_FabG). • Fed-batch fermentation revealed a 40% titer improvement (up to 6 g/L) in an optimized Al_FabG strain compared to a base strain and comparative proteomic data during fermentation revealed physiological changes related to NADPH availability . • Overall, the results suggest that heterologous expression of NADH-dependent FabG in E. coli may improve sustained production of fatty acid-derived renewable fuels and chemicals. Goh et al. (2018) Biotechnology and Bioengineering, doi: 10.1002/bit.26558/pdf Background • We previously engineered E. coli to overproduce medium- to long-chain saturated and monounsaturated methyl ketones, which could potentially be applied as diesel fuel blending agents or in the flavor and fragrance industry. • Fatty acid-derived pathways, such as methyl ketone biosynthesis, face the metabolic challenge of high NADPH demand, partly resulting from the key fatty acid biosynthetic enzyme, FabG (β-ketoacyl-ACP reductase). Approach • We attempted to mitigate cofactor imbalance by heterologously expressing NADH-dependent, rather than NADPH-dependent, versions of FabG that we identified in previous studies. • We evaluated the performance of engineered strains with fed-batch fermentation in 2-L reactors by measuring concentrations of methyl ketones and short-chain fatty acids, cell dry weight, and shotgun proteomic profiles. . Summary comparison of methyl ketone-producing strains of E. coli with and without expression of A. laidlawii FabG (Al_FabG). The Al_FabG strain showed higher methyl ketone titer and lower pyruvate accumulation (upper) and proteomic results at 50 hr showed differential expression of proteins associated with NADPH production. 0 2 4 6 8 10 0 1 2 3 4 5 6 0 50 100 150 200 250 OrganicacidsandCDW METHYLKETONES(g/L) Time (hr) MK with Al_FabG MK without Al_FabG FED-BATCH FERMENTATION 1.E-07 1.E-06 1.E-05 1.E-04 1.E-03 1.E-02 1.E-01 1.E+00 -10 -8 -6 -4 -2 0 2 4 6 8 p-value Log2 Fold Change PROTEOMICS @ 50 hr With Al_FabGWithout Al_FabG MaeB PGI
- 3. Identification of an algal xylan synthase indicates that there is functional orthology between algal and plant cell wall biosynthesis Background • Insights into the evolution of plant cell walls have important implications for comprehending these diverse and abundant biological structures. • In order to understand the evolving structure–function relationships of the plant cell wall, it is imperative to trace the origin of its different components. • 1,4-β-xylan is one of the most abundant components in plant biomass. Approach • The present study is focused on plant 1,4-β-xylan, tracing its evolutionary origin by genome and transcriptome mining followed by phylogenetic analysis, utilizing a large selection of plants and algae. It substantiates the findings by heterologous expression and biochemical characterization of an alga xylan synthase. Outcomes and Impacts • Of the 12 known gene classes involved in 1,4-β-xylan formation in plants, XYS1/IRX10, IRX7, IRX8, IRX9, IRX14 and GUX occurred for the first time in charophyte algae. An XYS1/IRX10 ortholog from Klebsormidium flaccidum, KfXYS1, possesses 1,4-β-xylan synthase activity, and 1,4-β-xylan occurs in the K. flaccidum cell wall. • These data suggest that plant 1,4-β-xylan originated in charophytes and shed light on the origin of one of the key cell wall innovations to occur in charophyte algae, facilitating terrestrialization and emergence of polysaccharide-based plant cell walls. Jensen et al. (2018) New Phytologist, doi: 10.1111/nph.15050 Phylogeny of the green lineage (Viridiplantae). The key genes required for synthesis of the hemicellulose 1,4-β- xylan evolved in a common ancestor of Klebsormidiophyceae and land plants. • Top: Enzymatic assay showing that KfXYS1 from Klebsormidium flaccidum has xylan synthase activity in vitro and can add xylose residues to a 1,4-β-xylotetraose acceptor substrate. Bottom: Cell walls from K. flaccidum contain 1,4-β-xylotetraose as showed by digestion with endo-xylanase and Polysaccharide Analysis using Carbohydrate gel Electrophoresis (PACE). Lane 2 is a no enzyme control, and the other lanes are combinations of xylanase and other enzymes. The pattern shows the presence of arabinofuranose substitutions and the absence of glycoronosyl substitutions.
- 4. Bi-functional glycosyltransferases catalyze both extension and termination of pectic galactan oligosaccharides Background • Pectins are the most complex polysaccharides of the plant cell wall. • Based on the number of methylations, acetylations, and glycosidic linkages present in their structures, it is estimated that up to 67 transferase activities are involved in pectin biosynthesis. • Pectic galactans constitute a major part of pectin in the form of side chains of rhamnogalacturonan-I. • In Arabidopsis, Galactan Synthase 1 (GALS1) catalyzes the addition of galactose units from UDP-Gal to growing β-1,4- galactan chains. • However, the mechanisms for obtaining varying degrees of polymerization remain poorly understood. Approach • In this study, we show that AtGALS1 is bifunctional, catalyzing both the transfer of galactose from UDP-α-D-Gal and the transfer of an arabinopyranose from UDP-β-L-Arap to galactan chains. • The two substrates share a similar structure, but UDP-α-D-Gal is the preferred substrate, with a tenfold higher affinity. • Transfer of Arap to galactan prevents further addition of galactose residues, resulting in a lower degree of polymerization. Outcomes and Impacts • We show that this dual activity occurs both in vitro and in vivo. • The herein described bi-functionality of AtGALS1 suggest that plants can produce the incredible structural diversity of polysaccharides without a dedicated glycosyltransferase for each glycosidic linkage. Laursen et al. (2018) Plant J., doi: 10.1111/tpj.13860. Schematic structure of pectic galactans. The GALS1 enzyme is responsible both for elongation of chains with galactose residues and for termination by arabinose residues. Enzyme assay showing the addition of galactose and arabinopyranose to galacto-pentaose by GALS1. Arabinofuranose is not transferred by GALS1. GALS1 was expressed in tobacco and a fluorescent labeled galactopentaose was used as acceptor substrate. Kinetic analysis of the transfer of galactose to galactopentaose by GALS1 shows a sigmoid curve indicating more than one substrate binding site.
- 5. Synthetic biology of polyketide synthases Background • Complex reduced polyketides represent the largest class of natural products that have applications in medicine, agriculture, and animal health, and are produced naturally by polyketide synthases (PKS). • We have used the nomenclature of synthetic biology to classify polyketide synthases as parts and devices, respectively, and have generated detailed lists of both. • Provides a common reference tool for the broader synthetic biology community. Approach • Developed a list of common synthetic biology syntax for PKSs – parts, devices and chassis. • Review of current approaches to engineer PKSs • Review of ClusterCAD, an online database to facilitate identification and selection for targeted PKSs for a particular pathway or pdoruct. Outcomes and Impacts • it should be possible to design a polyketide of desired structure, write out the list of parts and devices required, employ ClusterCAD or similar software to design the devices, synthesize the DNA of the required sequence, introduce the DNA into a desired chassis, and express it under a selected means of regulation. • The ability to link modules in sequence to generate completely new polyketide structures without losing biochemical activity is still extremely challenging. Yuzawa et al. (2018) Ind Microbiol Biotechnol., doi: 10.1007/s10295-018-2021-9 (A) Example chemical target used to demonstrate guiding chimeric PKS engineering with the ClusterCAD software tool. (B) A maximum common substructure (MCS) plot highlighting the common regions between the example chemical target (on right), and narbonolide, a chemically similar natural PKS found in ClusterCAD (on left). The non-highlighted regions represent engineering modifications necessary to produce the example product. Structures of malonyl-CoA analogs that are incorporated during biosynthesis of naturally occurring polyketides.
- 6. Outcomes and Impacts • A mixture of 42 wt.% [C2C1Im][OAc] and 58 wt.% DMSO is proposed as the optimal pretreatment solution and the recycling and reuse of the mixture of solvents are also studied. • The fermentability of the hydrolysates generated after pretreatment is evaluated using an E. coli strain engineered to produce isoprenol, and the highest isoprenol yields generated were 24.5 g/kg switchgrass. • This study suggests an avenue for developing more efficient and cost-effective IL-based processes for the production of biofuels and bioproducts. Wang et al. (2018) ACS Sustainable Chem. Eng., doi: 10.1021/acssuschemeng.7b04908 Background • The production cost and viscosity of certain ionic liquids (ILs) are among the major factors preventing the establishment of economically viable IL-based biomass pretreatment technologies. Approach • One of the approaches to reduce the cost of IL pretreatment has been the use of mixtures of ILs with inexpensive solvents such as dimethyl sulfoxide (DMSO) • We conducted a mechanistic study of DMSO-assisted IL pretreatment of switchgrass • An engineered E. coli strain, KG1R10, containing a heterologous mevalonate-based isoprenol pathway was used for isoprene production. Dimethyl sulfoxide assisted ionic liquid pretreatment of switchgrass for isoprenol production