JBEI Highlights May 2015
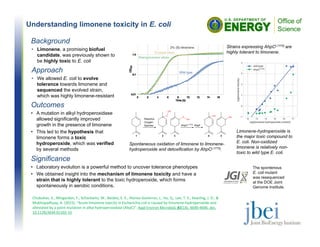
- 1. Understanding limonene toxicity in E. coli Outcomes • A mutation in alkyl hydroperoxidase allowed significantly improved growth in the presence of limonene • This led to the hypothesis that limonene forms a toxic hydroperoxide, which was verified by several methods Background • Limonene, a promising biofuel candidate, was previously shown to be highly toxic to E. coli Approach • We allowed E. coli to evolve tolerance towards limonene and sequenced the evolved strain, which was highly limonene-resistant Significance • Laboratory evolution is a powerful method to uncover tolerance phenotypes • We obtained insight into the mechanism of limonene toxicity and have a strain that is highly tolerant to the toxic hydroperoxide, which forms spontaneously in aerobic conditions. Spontaneous oxidation of limonene to limonene- hydroperoxide and detoxification by AhpCL177Q. Chubukov, V., Mingardon, F., Schackwitz, W., Baidoo, E. E., Alonso‐Gutierrez, J., Hu, Q., Lee, T. S., Keasling, J. D., & Mukhopadhyay, A. (2015). "Acute limonene toxicity in Escherichia coli is caused by limonene‐hydroperoxide and alleviated by a point mutation in alkyl hydroperoxidase (AhpC)". Appl Environ Microbiol, 81(14), 4690‐4696. doi, 10.1128/AEM.01102‐15 Strains expressing AhpCL177Q are highly tolerant to limonene. Limonene-hydroperoxide is the major toxic compound to E. coli. Non-oxidized limonene is relatively non- toxic to wild type E. coli. The spontenous E. coli mutant was resequenced at the DOE Joint Genome Institute.
- 2. Metabolic engineering for the high-yield production of isoprenoid-based C5 alcohols in E. coli Outcomes • 60% increase in the yield of 3-methyl-3-buten-1-ol by engineering the Shine-Dalgarno sequence of nudB2 • Achieved final titers of 2.23 g/L of 3-methyl-3-buten-1-ol (~70% of pathway-dependent theoretical yield)2 1George, et al., “Correlation Analysis of Targeted Proteins and Metabolites to Assess and Engineer Microbial Isopentenol Production.” Biotechnology and Bioengineering. 111(8):1648-1658 (2014). 2George, et al., “Metabolic engineering for the high-yield production of isoprenoid-based C5 alcohols in E. coli.” Scientific Reports. doi:10.1038/srep11128 (2015). Background • Branched five carbon (C5) alcohols are attractive targets for microbial production due to their desirable fuel properties and importance as platform chemicals • Optimization of Initially engineered two-plasmid system for 3-methyl-3- buten-1-ol has been performed1 Approach • NudB, a promiscuous phosphatase, was identified as a likely pathway bottleneck by metabolite profiling, and RBS engineering on NudB was attempted. • Further engineering on mevalonate kinase expression and C5 alcohol recovery was also attempted. Significance • Engineered a heterologous isoprenoid pathway in E. coli for the high-yield production of 3-methyl-3-buten-1-ol, 3-methyl-2-buten-1-ol, and 3-methyl-1-butanol RBS engineering to improve 3-methyl-3-buten-1-ol titer and reduce IPP accumulation. ~70% theoretical yield and 2.2 g/L titer in shake flask w/ overlay
- 3. Engineering of plant cell walls for enhanced biofuel production 1Linshiz, et al., “PaR-PaR: Laboratory Automation System.” ACS Synth. Biol. 2:216-222 (2013). 2Linshiz, et al., “PR-PR: Cross-Platform Laboratory System.” ACS Synth. Biol. Article ASAP (2014). Background • In this publication, we provide an overview of current advances in engineering of plants with improved properties as feedstocks for biofuels production. • The review focused on the following targets: • Reducing lignin • Decreasing inhibitors such as acetate • Increasing hexose/pentose ratio • Increasing cell wall sugar content Significance/perspective • Up to date review on the advances in cell wall engineering. • Summary of many recent studies including several from the BRCs. • Annotated reference list with 87 references to mostly recent literature • Discussion of remaining challenges, especially trait stacking and predicting plant performance under a range of environmental conditions. Loque D, Scheller HV, Pauly M (2015) Engineering of plant cell walls for enhanced biofuel production. Curr Opin Plant Biol 25: 151‐161. An example of engineering to obtain high density and low lignin. In these plants, normal lignin was maintained in the vessels while low lignin was restricted to fiber cells. Biomass is composed of different polysaccharides and lignin. The detailed composition differs between different types of feedstock.
- 4. An unusual xylan in Arabidopsis primary cell walls is synthesised by GUX3, IRX9L, IRX10L and IRX14 Background • The polysaccharide xylan is a major component of biomass (2nd only to cellulose). • It is a hindrance to cellulose deconstruction (blocks cellulase access); difficult to depolymerise. Approach and Outcomes • We identified and characterized a novel xylan structure in Arabidopsis. This xylan had glycosidic linkages resistant to standard xylan degrading hydrolases. • This xylan also has an novel pattern of side-chain spacing, pointing to exquisite molecular control over glycan synthesis. This pattern has unknown function. • Using a multi ‘omics approach, we identified candidate glycosyltransferase genes for its biosynthesis. • We used reverse genetics to test and confirm these predictions in planta. • We developed a high-throughput, non-radioactive methods for characterizing glycosyltransferase activity in vitro. Significance • We characterized a novel xylan structure and identified the majority of the genes responsible for its synthesis. Mortimer, JC et al. (2015). An unusual xylan in Arabidopsis primary cell walls is synthesised by GUX3, IRX9L, IRX10L and IRX14. Plant Journal. doi, 10.1111/tpj.12898 Cellulose Xylan Work led by Prof. Paul Dupree, University of Cambridge, UK.
- 5. Trends In Microbiology: engineering solvent tolerant microbes The review I reviewed strain engineering, primarily as it pertains to bacterial solvent tolerance, and on the benefits and challenges associated with expression of membrane‐ localized transporters in improving solvent tolerance and production. Background During microbial production of solvent‐like compounds, such as advanced biofuels and bulk‐chemicals, accumulation of the final product can negatively impact the cultivation of the host microbe and limit the production levels. Consequently, improving solvent‐ tolerance is becoming an essential aspect of engineering microbial production strains. Transporters specifically have emerged as a powerful category of proteins that bestow tolerance and often improve production but are difficult targets for cellular expression. Mukhopadhyay, A. (2015). "Tolerance engineering in bacteria for the production of advanced biofuels and chemicals". Trends Microbiol. doi, 10.1016/j.tim.2015.04.008
- 6. SRM-based approach to assess organelle profiles in plant samples Outcomes • It is indeed possible to use the SRM approach (targeted mass spectrometry) to profile organelle abundance in a plant sample • We also demonstrated that the approach can be used to assess organelle profiles in plant tissues e.g. the abundance of plastid markers was significantly higher in photosynthetic tissue. Parsons and Heazlewood (2015). Beyond the Western front: targeted proteomics and organelle abundance profiling. Front. Plant Sci. 5, 301. doi: 10.3389/fpls.2015.00301 Background • The characterization of subcellular organelles involved in cell wall biosynthesis requires high purity preparations for proper analyses. • Although western blotting enables profiling of organelle purity and contamination, there is a limited availability of antibodies in plant science Approach • Use targeted mass spectrometry approaches to profile protein extracts to determine suitability of this method for organelle profiling. Significance • We have developed a collection of SRM transitions or peptide targets that can be now be used to easily and quickly assess plant organelle profiles in fractions associated with cell wall biosynthesis e.g. plasma membrane and Golgi apparatus. Non‐photosynthetic tissue Photosynthetic tissue SRM signal for plastid markers from mass spectrometry 1 2 3 1 2 3
- 7. Complex regulation of prolyl-4-hydroxylases impacts root hair expansion 1Linshiz, et al., “PaR-PaR: Laboratory Automation System.” ACS Synth. Biol. 2:216-222 (2013). 2Linshiz, et al., “PR-PR: Cross-Platform Laboratory System.” ACS Synth. Biol. Article ASAP (2014). Background • Prolyl-4-hyroxylase is an enzyme responsible for converting proline to hydroxyproline. The Hydroxyproline residues are subsequently glycosylated. Thus, this step is essential for formation of many, possibly all, glycans and polysaccharies in the plant cell wall. • Plants have several P4H isoforms and their individual roles have not been understood. Approach and Outcomes • Mutants in three major P4H isoforms expressed in Arabidopsis roots were studied. • P4H5 has an essential function different from P4H2 and P4H13, which are partly redundant. • Protein-protein interactions were investigated by co- localization, bimolecular fluorescence complementation (BiFC) and FRET (Förster resonance energy transfer). • The P4H proteins form homodimers, and additionally forms heterodimes with P4H5 • P4H5 is localized in the Er when P4H5 is absent. Only in the presence of P4H5 is the P4H2 protein targeted to Golgi. Significance • Understanding P4H function is important since these enzymes are necessary for glycan formation in plant cell walls. The conditional targeting of P4H2 is an important illustration of the role of protein- protein interactions in the localization of function fo Golgi resident proteins involved in cell wall formation. This is a vastly understudied area of research. Velasquez al. (2015). "Complex regulation of prolyl‐4‐hydroxylases impacts root hair expansion". Mol Plant 8(5), 734‐746. Left: In roots, deficiency in P4H is seen in the absence or deficiency in root hairs. Below: Protein‐protein interactions were investigated with a range of methods. Results obtained with BiFC are shown.