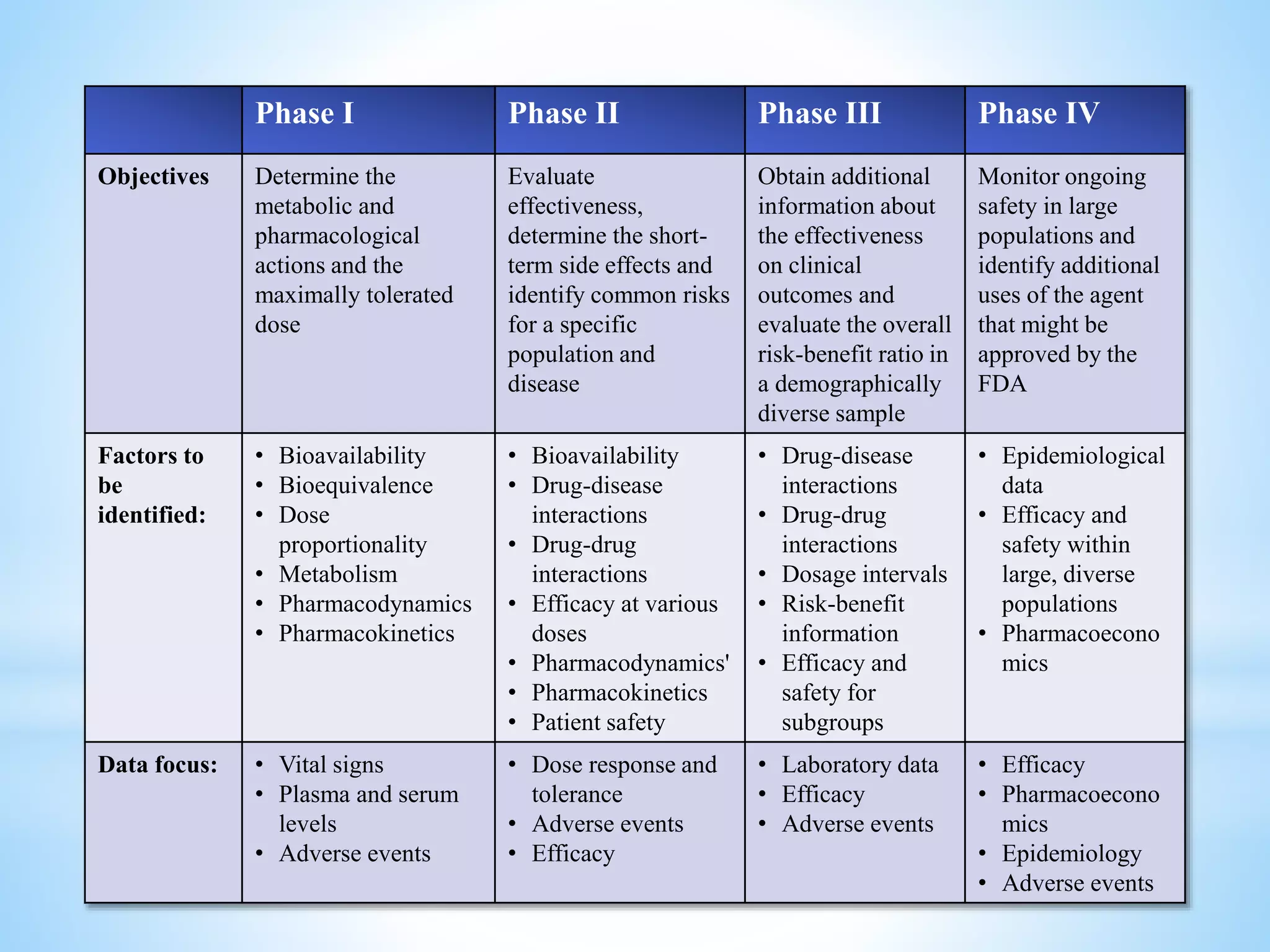
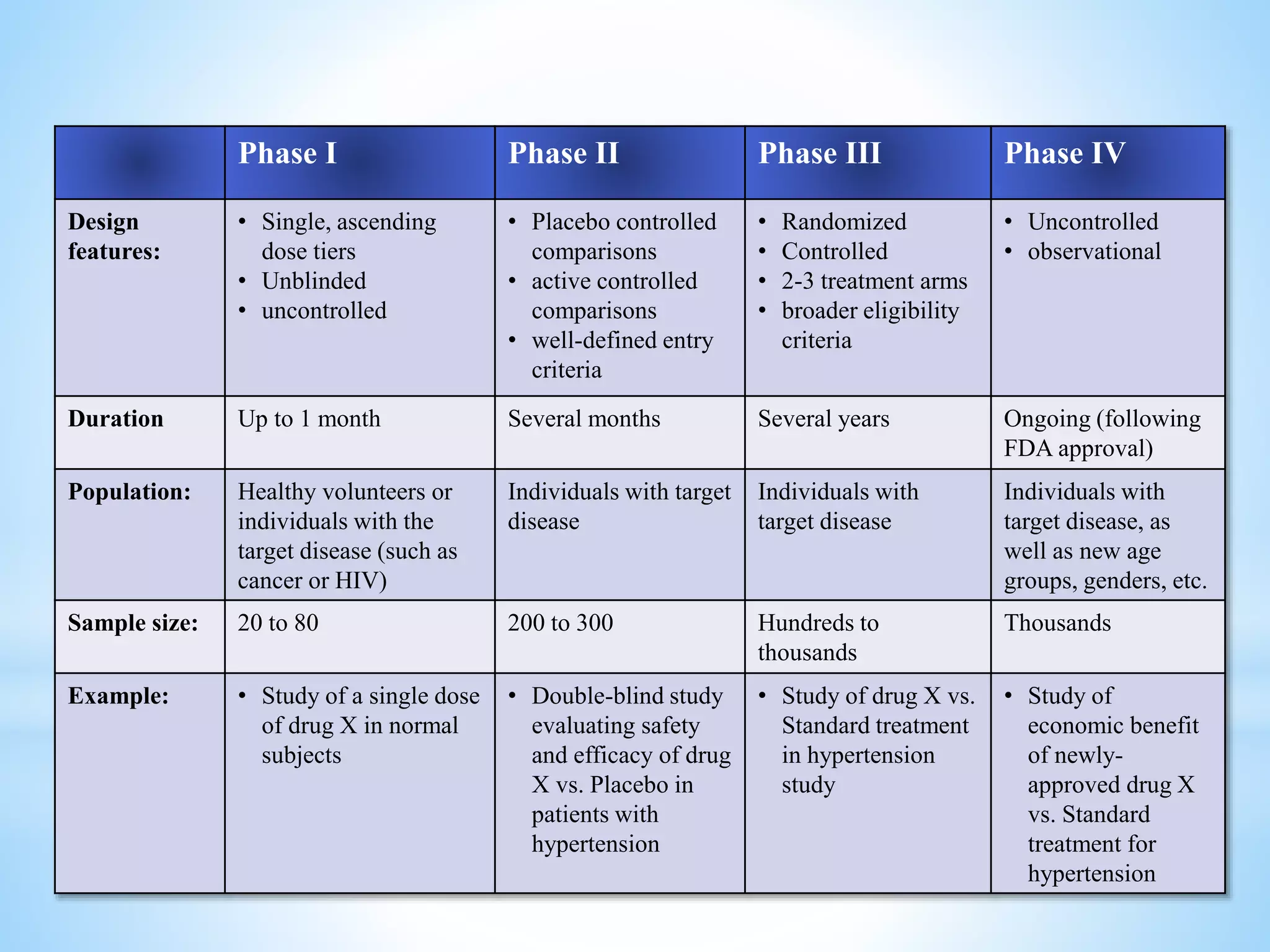
The document provides a comprehensive overview of clinical trials, outlining their history, types, and designs, including observational and experimental approaches. It describes key phases of clinical trials, factors to be considered, and various study designs such as randomized controlled trials, case-control studies, and cohort studies. Additionally, the document discusses the importance of randomization, potential biases, and ethical considerations in conducting clinical research.