Solventextraction 140711154150-phpapp02
•Download as PPT, PDF•
0 likes•25 views
1) Solvent extraction is a technique used to separate components in a mixture based on differences in solubility between two immiscible liquid phases. 2) It involves transferring a solute from one liquid phase to another, such as transferring a compound from an aqueous phase to an organic phase like benzene. 3) The amount of solute extracted into each phase can be calculated using the partition coefficient K, which is a ratio of concentrations in the two phases at equilibrium.
Report
Share
Report
Share
Recommended
More Related Content
What's hot
What's hot (20)
Report on pH and conductivity of different samples from delhi

Report on pH and conductivity of different samples from delhi
Aerobic Biodegradation of Vinegar Containing Waste Water by Mixed Culture Bac...

Aerobic Biodegradation of Vinegar Containing Waste Water by Mixed Culture Bac...
Similar to Solventextraction 140711154150-phpapp02
Similar to Solventextraction 140711154150-phpapp02 (11)
Poster-Next Generation Self-Healing Concrete-Infusing Bacteria into Engineere...

Poster-Next Generation Self-Healing Concrete-Infusing Bacteria into Engineere...
Determination of the molecular weight of high polyvinyl alcohol (pva) by visc...

Determination of the molecular weight of high polyvinyl alcohol (pva) by visc...
Synthesis of E, E-dibenzalacetone (E, E-DBA) Lab ReportN.docx

Synthesis of E, E-dibenzalacetone (E, E-DBA) Lab ReportN.docx
More from Cleophas Rwemera
More from Cleophas Rwemera (20)
Chapter22 themolecularcellbiologyofdevelopment-140105100412-phpapp02

Chapter22 themolecularcellbiologyofdevelopment-140105100412-phpapp02
Chapter21 cellbirthlineageanddeath-140105095914-phpapp02

Chapter21 cellbirthlineageanddeath-140105095914-phpapp02
Chapter20 regulatingtheeukaryoticcellcycle-140105095738-phpapp01

Chapter20 regulatingtheeukaryoticcellcycle-140105095738-phpapp01
Chapter19 integratingcellsintotissues-140105095535-phpapp02

Chapter19 integratingcellsintotissues-140105095535-phpapp02
Chapter18 cellorganizationandmovementiimicrotubulesandintermediatefilaments-1...

Chapter18 cellorganizationandmovementiimicrotubulesandintermediatefilaments-1...
Chapter17 cellorganizationandmovementimicrofilaments-140105094810-phpapp02

Chapter17 cellorganizationandmovementimicrofilaments-140105094810-phpapp02
Chapter16 cellsignalingiisignalingpathwaysthatcontrolgeneactivity-14010509451...

Chapter16 cellsignalingiisignalingpathwaysthatcontrolgeneactivity-14010509451...
Chapter15 cellsignalingisignaltransductionandshort-termcellularresponses-1401...

Chapter15 cellsignalingisignaltransductionandshort-termcellularresponses-1401...
Chapter14 vesiculartrafficsecretionandendocytosis-140105094215-phpapp01

Chapter14 vesiculartrafficsecretionandendocytosis-140105094215-phpapp01
Chapter13 movingproteinsintomembranesandorganelles-140105094005-phpapp01

Chapter13 movingproteinsintomembranesandorganelles-140105094005-phpapp01
Chapter12 cellularenergetics-140105093734-phpapp01

Chapter12 cellularenergetics-140105093734-phpapp01
Chapter11 transmembranetransportofionsandsmallmolecules-140105092904-phpapp02

Chapter11 transmembranetransportofionsandsmallmolecules-140105092904-phpapp02
Chapter10 biomembranestructure-140105093829-phpapp02

Chapter10 biomembranestructure-140105093829-phpapp02
Chapter9 visualizingfractionatingandculturingcells-140105092245-phpapp01

Chapter9 visualizingfractionatingandculturingcells-140105092245-phpapp01
Recently uploaded
Operations Management - Book1.p - Dr. Abdulfatah A. Salem

Operations Management - Book1.p - Dr. Abdulfatah A. SalemArab Academy for Science, Technology and Maritime Transport
Recently uploaded (20)
Incoming and Outgoing Shipments in 2 STEPS Using Odoo 17

Incoming and Outgoing Shipments in 2 STEPS Using Odoo 17
Matatag-Curriculum and the 21st Century Skills Presentation.pptx

Matatag-Curriculum and the 21st Century Skills Presentation.pptx
The impact of social media on mental health and well-being has been a topic o...

The impact of social media on mental health and well-being has been a topic o...
slides CapTechTalks Webinar May 2024 Alexander Perry.pptx

slides CapTechTalks Webinar May 2024 Alexander Perry.pptx
Salient features of Environment protection Act 1986.pptx

Salient features of Environment protection Act 1986.pptx
aaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaa

aaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaaa
Benefits and Challenges of Using Open Educational Resources

Benefits and Challenges of Using Open Educational Resources
Operations Management - Book1.p - Dr. Abdulfatah A. Salem

Operations Management - Book1.p - Dr. Abdulfatah A. Salem
Solventextraction 140711154150-phpapp02
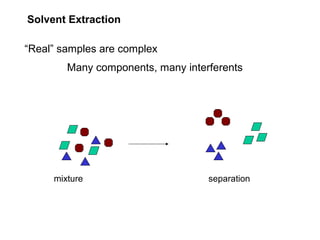
- 1. Solvent Extraction “Real” samples are complex Many components, many interferents mixture separation
- 2. What to do: a. Masking agents b. Precipitation and Filtration c. Distillation d. Extraction e. Chromatography
- 4. Solvent Extraction Isolate individual components based on differences in solubility, pH… Transfer a solute from one phase to another Phase 1 (benzene) Phase 2 (water) Aaq = Aorg Activity (A) org Activity (A) aq K = Partition Coeff
- 5. To calculate how much is in each phase, K Volume of organic phase Volume of aqueous phase Initial concentration in aqueous phase n orgaq aq KVV V q )( + = [ ] ( ) [ ]A V V V K Aaq n aq aq org n aq= + 0 n = # times
- 8. Much better to use a large # of small portions of solvent, rather than one large one Fraction remaining # extractions
- 9. It is desired to extract 80.0% of the amylacetate from a banana peel in one extraction. If K is 10 and the volume of water the banana peel is homogenized in is 50.0 mL, how many mL of organic solvent will be required. a. 1.25 b. 20 c. 50 d. 65 Clicker question:
- 10. It is desired to extract 80.0% of the amylacetate from a banana peel in one extraction. If K is 10 and the volume of water the banana peel is homogenized in is 50.0 mL, how many mL of organic solvent will be required. a. 1.25 b. 20 c. 50 d. 65 Clicker question: