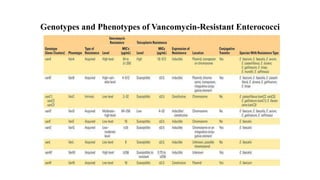
Vancomycin-resistant enterococci (VRE) are a significant cause of hospital-acquired infections. Enterococcus faecium and Enterococcus faecalis are the most common clinical species. VRE have acquired resistance to vancomycin through the alteration of peptidoglycan precursors in cell wall synthesis. The vanA and vanB genotypes are most prevalent and confer high-level resistance. Treatment involves combination therapy with cell wall active agents and aminoglycosides. Early detection of VRE enables timely infection control measures to limit spread.