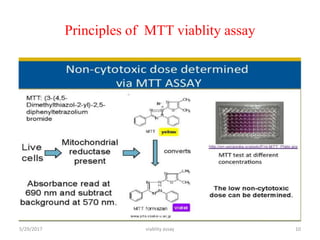
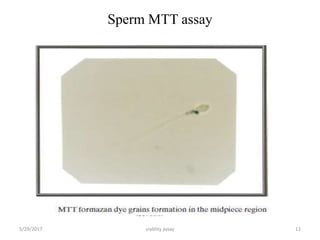
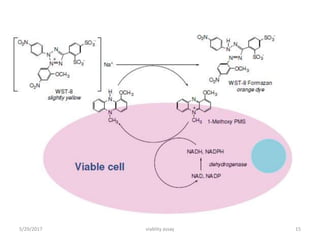
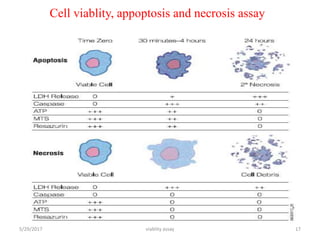
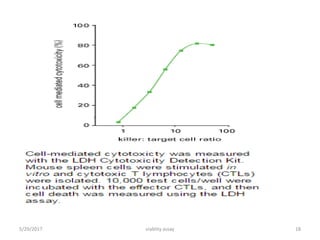
This document discusses viability assays, which determine if cells are alive or dead. It defines viability assays and explains they are based on cell functions like enzyme activity and membrane permeability. Common viability assays described include MTT, WST, trypan blue, and LDH assays. The MTT and WST assays work by measuring the reduction of tetrazolium salts by metabolically active cells, indicating the number of living cells. Trypan blue distinguishes live from dead cells by staining only dead cells blue. The document classifies viability assays and explains their applications in screening drugs, diagnosing diseases, and measuring cell survival.