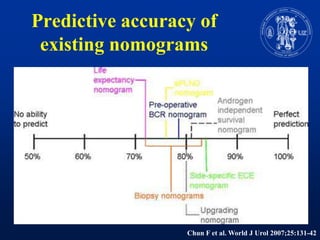
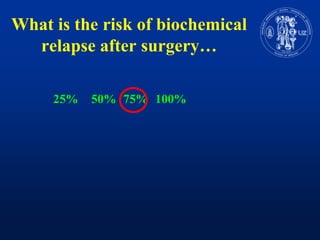
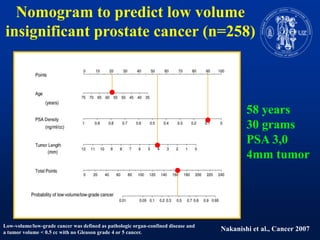
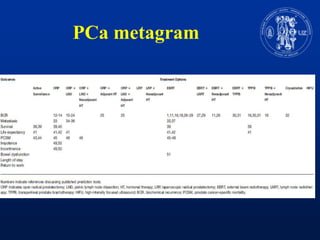
Nomograms provide predictions of outcomes for prostate cancer patients based on known treatment outcomes of similar patients. However, nomograms have several limitations including bias from the development cohort, lack of external validation, and lack of updates using contemporary patient populations. Additionally, nomograms often use surrogate endpoints rather than clinically meaningful endpoints and predictive accuracy is not 100%. While nomograms can help guide clinical decision making, good clinical judgement is still needed and nomograms may not accurately capture all risk factors or change clinical decisions for individual patients.