Science and Stories: Navigating the Prostate Cancer Landscape - Urologists at the Intersection of Emerging Evidence and Patient Centric Care
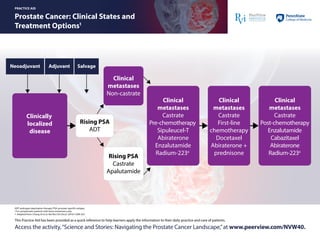
- 1. ADT: androgen deprivation therapy; PSA: prostate-specific antigen. a For symptomatic patients with bone metastases only. 1. Adapted from: Chang JA et al. Nat Rev Clin Oncol. 2014;11:308-323. Prostate Cancer: Clinical States and Treatment Options1 PRACTICE AID This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. Access the activity,“Science and Stories: Navigating the Prostate Cancer Landscape,”at www.peerview.com/NVW40. Neoadjuvant Clinically localized disease Clinical metastases Castrate Pre-chemotherapy Sipuleucel-T Abiraterone Enzalutamide Radium-223a Clinical metastases Castrate Post-chemotherapy Enzalutamide Cabazitaxel Abiraterone Radium-223a Clinical metastases Castrate First-line chemotherapy Docetaxel Abiraterone + prednisone Clinical metastases Non-castrate Rising PSA ADT Rising PSA Castrate Apalutamide Adjuvant Salvage
- 2. Key Ongoing Phase 3 Clinical Trials in Prostate Cancer1 PRACTICE AID This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. Localized or Locally Advanced Prostate Cancer ATLAS (NCT02531516) GnRH agonist + apalutamide vs GnRH agonist + bicalutamide High-risk patients receiving primary RT ECOG PS 0 or 1 Metastasis-free survival OS Active Not recruiting Planned N = 350 Recruiting Planned N = 1500 Non-Metastatic CRPC Metastasis-free survival Active Has results N = 1207 Metastasis-free survival Active Has results N = 1401 Metastasis-free survival EMBARK (NCT02319837) Enzalutamide + leuprolide vs enzalutamide vs placebo + leuprolide High-risk hormone-sensitive patients progressing after definitive therapy (radical prostatectomy or radiotherapy or both) Metastasis-free survival Recruiting Planned N = 1860 Treatment and population Treatment and population Primary endpoint Primary endpoint Active Not recruiting Planned N = 1502 SPARTAN (NCT01946204) Apalutamide vs placebo High-risk castration-resistant patients ECOG PS 0 or 1 PROSPER (NCT02003924) Enzalutamide vs placebo Ongoing ADT or prior bilateral orchiectomy ECOG PS 0 or 1 ARAMIS (NCT02200614) Darolutamide vs placebo High-risk castration-resistant patients ECOG PS 0 or 1 LoLA PC (NCT00116142) Androgen suppression + RT with or without docetaxel High-risk patients ECOG PS 0 or 1 Access the activity,“Science and Stories: Navigating the Prostate Cancer Landscape,”at www.peerview.com/NVW40.
- 3. Key Ongoing Phase 3 Clinical Trials in Prostate Cancer1 PRACTICE AID This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. Metastatic Hormone-Sensitive Prostate Cancer TITAN (NCT02489318) ADT + placebo vs ADT + apalutamide Newly-diagnosed or previously treated ECOG PS 0 or 1 OS and radiographic PFS Active Not recruiting Planned N = 1052 ARASENS (NCT02799602) ADT + docetaxel + placebo vs ADT + darolutamide + docetaxel Newly-diagnosed metastatic disease ECOG PS 0 or 1 OS Recruiting Planned N = 1300 OS and PFS Recruiting Planned N = 1168 LATITUDE (NCT01715285) Abiraterone + low-dose prednisone + ADT vs ADT alone High-risk patients ECOG PS 0-2 OS and radiographic PFS Active Has results N = 1199 Radiographic PFS Active Not recruiting Planned N = 1150 Treatment and population Treatment and population Primary endpoint Primary endpoint ARCHES (NCT02677896) ADT + enzalutamide vs ADT + placebo Ongoing ADT or prior bilateral orchiectomy ECOG PS 0 or 1 PEACE1 (NCT01957436) ADT + docetaxel +/- local RT +/- abiraterone ECOG PS 0 or 1 Access the activity,“Science and Stories: Navigating the Prostate Cancer Landscape,”at www.peerview.com/NVW40.
- 4. ADT: androgen deprivation therapy; AR: androgen receptor; CRPC: castrate-resistant prostate cancer; ECOG: Eastern Cooperative Oncology Group; GnRH: gonadotropin-releasing hormone agonist; OS: overall survival; PFS: progression-free survival; PS: performance status; RT: radiotherapy; vs: versus. 1. http://www.clinicaltrials.gov. Accessed April 23, 2018. Key Ongoing Phase 3 Clinical Trials in Prostate Cancer1 PRACTICE AID This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. Access the activity,“Science and Stories: Navigating the Prostate Cancer Landscape,”at www.peerview.com/NVW40. Metastatic CRPC Radiographic PFS NCT02257736 Apalutamide + abiraterone + prednisone vs abiraterone + prednisone Chemotherapy-naïve patients Radiographic PFS Recruiting Planned N = 400 ALLIANCE A031201 (NCT01949337) Enzalutamide + abiraterone + prednisone vs enzalutamide alone Ongoing ADT or prior bilateral orchiectomy ECOG PS 0 or 1 OS Active Not recruiting Planned N = 1311 Active Not recruiting Planned N = 983 IPATential150 (NCT03072238) Ipatasertib + abiraterone + prednisone/prednisolone vs placebo + abiraterone + prednisone/prednisolone Previously untreated, asymptomatic or mildly symptomaticdisease;ongoing ADTorpriorbilateralorchiectomy ECOG PS 0 or 1 Radiographic PFS PROfound Study (NCT02987543) Olaparib vs enzalutamide or abiraterone Previously treated with abiraterone and/or enzalutamide; ongoing ADT or prior bilateral orchiectomy Radiographic PFS ERA 223 (NCT02043678) Radium-223 dichloride + abiraterone + prednisone/ prednisolone vs placebo + abiraterone + prednisone/ prednisolone Asymptomatic or mildly symptomatic; chemotherapy-naïve bone-predominant disease ECOG PS 0 or 1 Symptomatic skeletal event-free survival Active Not recruiting Planned N = 806 Treatment and population Treatment and population Primary endpoint Primary endpoint Recruiting Planned N = 850 Recruiting Planned N = 340 TRITON3 (NCT02975934) Rucaparib vs physician's choice of therapy Patients with deleterious mutation in a BRCA1/2 or ATM gene; 1 prior next generation AR-targeted therapy
