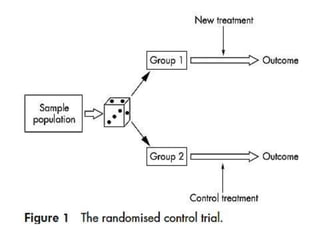
Randomization aims to equally distribute participant characteristics between treatment groups to prevent bias. There are several types of randomization including simple, block, and stratified block randomization. Blinding, such as double or triple blinding, helps prevent performance, detection, and other biases by keeping parties unaware of treatment assignments. Bias can still occur through factors like selection, performance, detection, laboratory, or sample size biases if randomization and blinding are not properly implemented.