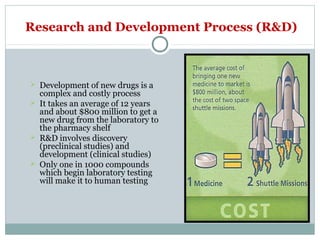
The document outlines the drug registration process in the U.S., detailing the stages of drug discovery, development, and FDA approval, which typically takes 12 to 15 years and $800 million. It discusses the complexities of clinical trials, emphasizing that only a small fraction of compounds advance to human testing and eventual market approval. Key roles of the FDA in evaluating drug safety and effectiveness through various phases of clinical studies are highlighted, along with the regulatory framework established throughout U.S. history.