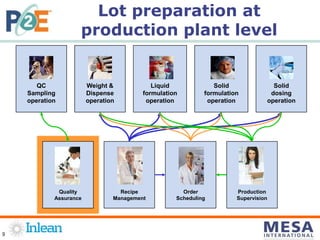
This document summarizes a presentation on improving operations performance through electronic guides (eGuides) in regulated industries. It discusses how traditional manufacturing documentation is often paper-based, including standard operating procedures, work orders, and quality records. An eGuide moves this documentation to a digital system, with contents and documents converted to digital files interconnected in a plant operations model. Implementing an eGuide provides benefits like centralized access to documents, version control, electronic signatures, and improved traceability and reporting. The presentation provides examples of how eGuides can model production processes and guide operators through work orders. It also discusses resources for defining, approving, executing, tracking, and analyzing manufacturing operations using eGuides.