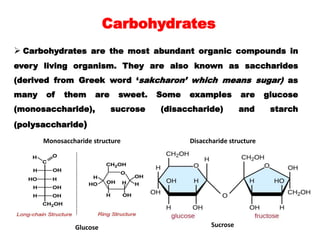
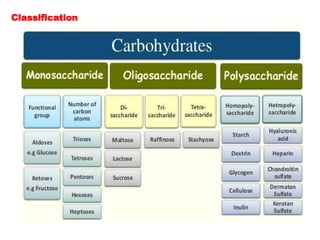
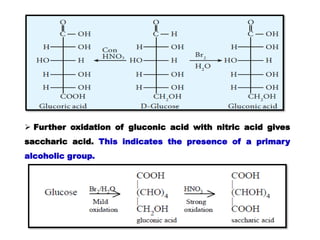
The document provides an overview of the structure of glucose, a monosaccharide carbohydrate. It discusses the classification of carbohydrates and describes experiments that were conducted to determine glucose's molecular formula and structure, including elemental analysis, reactions with various reagents, and oxidation reactions. The structure of glucose is shown to be an open-chain aldehyde with five hydroxyl groups and the molecular formula C6H12O6.
![Assistant Professor
Department of Chemistry
Faculty of Science
SCSVMV
[Deemed to be university]
Enathur, Kanchipuram, Tamilnadu
Dr. B. MUTHUKUMARAN
ORGANIC CHEMISTRY-II
CARBOHYDRATE-
STRUCTURE OF GLUCOSE](https://image.slidesharecdn.com/structureofglucose-220427052803/75/Structure-of-glucose-pdf-1-2048.jpg)