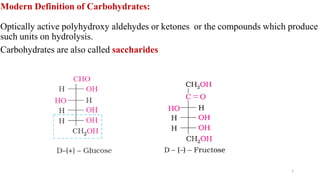
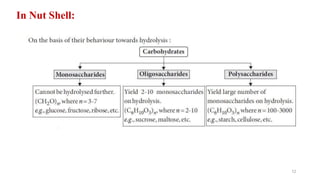
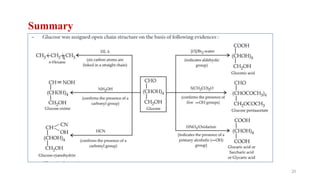
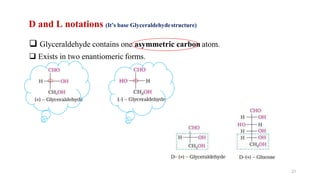
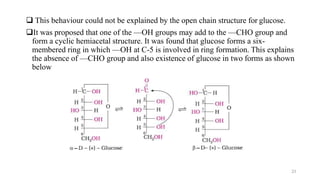
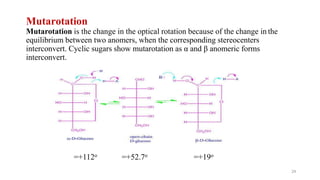
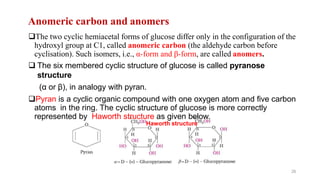
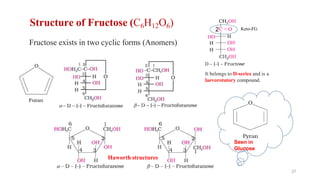
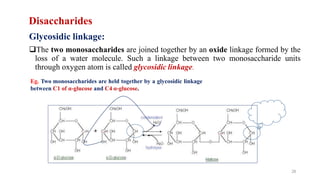
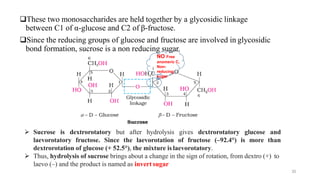
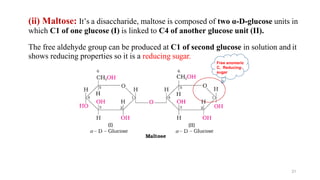
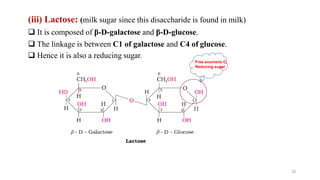
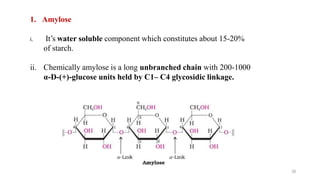
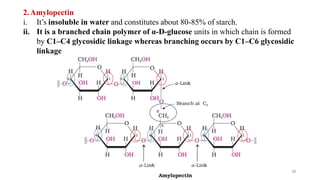
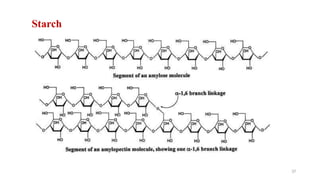
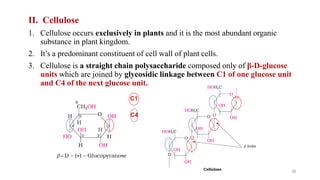
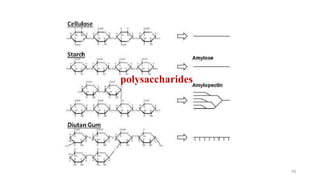
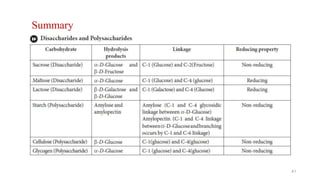
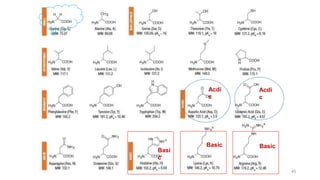
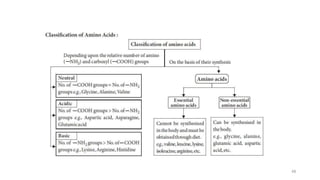
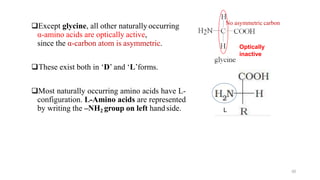
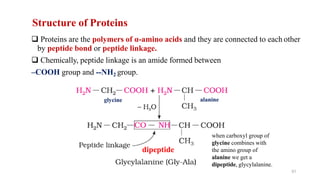
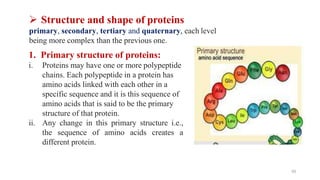
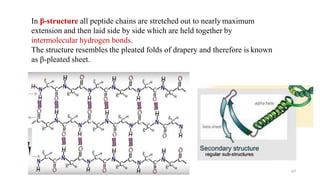
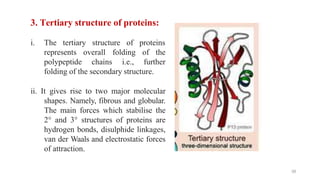
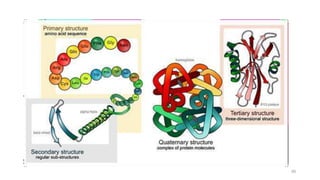
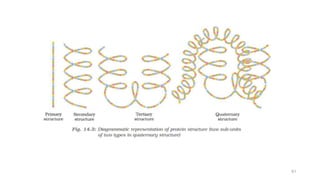
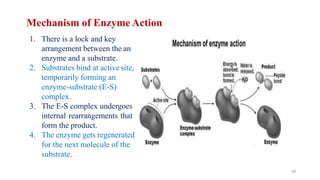
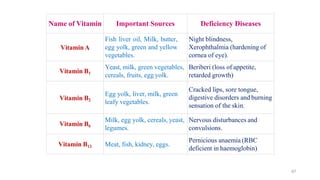
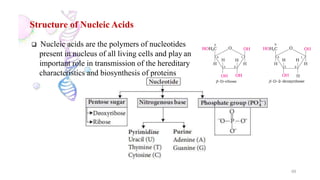
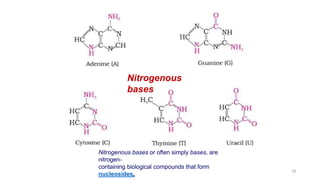
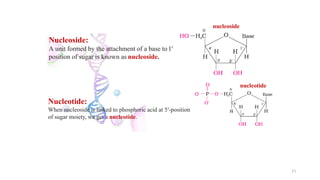
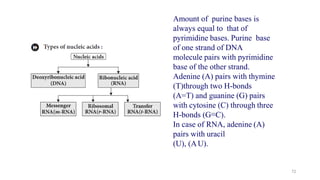
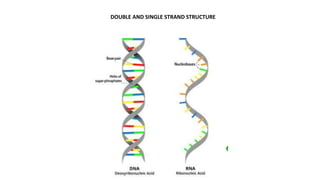
The document discusses different types of biomolecules found in the human body, focusing on carbohydrates. It defines carbohydrates and classifies them into monosaccharides, oligosaccharides, and polysaccharides based on their structure. Common carbohydrates like glucose, fructose, and sucrose are described in detail. The cyclic and linear structures of glucose are explained. Carbohydrates serve important functions as an energy source and building blocks in the body.