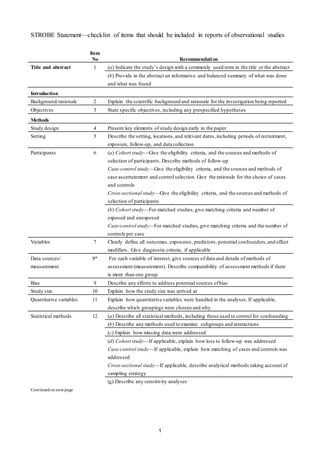
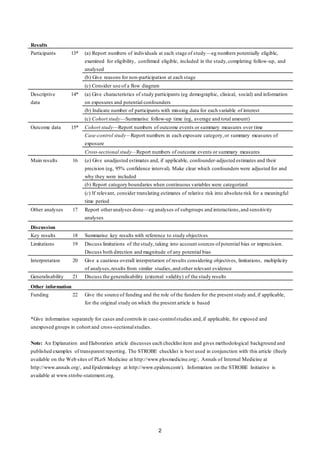
This document outlines 22 checklist items that should be included when reporting observational studies. It includes recommendations for what to report in the title/abstract, introduction, methods, results, and discussion sections. Some key recommendations include indicating the study design in the title or abstract, stating the specific objectives and hypotheses, describing the study setting and dates, defining all variables, describing statistical methods used, and discussing limitations and generalizability. Providing these details helps ensure transparent and complete reporting of observational studies.