Strobe checklist cross-sectional
•Download as DOC, PDF•
0 likes•225 views
This document provides a checklist of 22 items that should be included when reporting the results of cross-sectional studies. The checklist includes recommendations for the title, abstract, introduction, methods, results, discussion, and other sections of reports. Key elements that should be reported include the study design, setting, participants, variables, data sources, quantitative handling, statistical methods, numbers of participants, descriptive data, outcome data, estimates, subgroups, limitations, interpretation, and funding sources. Use of the checklist aims to improve transparent reporting of cross-sectional studies.
Report
Share
Report
Share
Recommended
NCD Risk factor Surveillance

This presentation covers risk factor assessment and surveillance of Non-communicable diseases.
International health regulations(ihr).pptx

international health regulations
by vibham gulati and sahil mogla (MBBS)
adesh univeristy
Rntcp and national strategic plan(nsp) for tb

After the successful NSP 2017-2025,Goi is lauching NSP 2017-2025 for elimination of TB on 24th march( World TB day ) 2017. Module is on MOHFW site but i have try to keep it brief,hope its ll be useful specially for academic and administrative purposes.
Recommended
NCD Risk factor Surveillance

This presentation covers risk factor assessment and surveillance of Non-communicable diseases.
International health regulations(ihr).pptx

international health regulations
by vibham gulati and sahil mogla (MBBS)
adesh univeristy
Rntcp and national strategic plan(nsp) for tb

After the successful NSP 2017-2025,Goi is lauching NSP 2017-2025 for elimination of TB on 24th march( World TB day ) 2017. Module is on MOHFW site but i have try to keep it brief,hope its ll be useful specially for academic and administrative purposes.
Public health emergencies

India being a developing country with growing population has been traditionally vulnerable to natural and man made disasters.
Development cannot be sustainable unless disaster mitigation is built into developmental process.
Disaster could be a nature calamity, outbreak of disease, bioterrorism, etc.
New Delhi, Feb 23. The health ministry has proposed a bill that seeks to empower state and local authorities to take appropriate actions to tackle public health emergencies like epidemics and bio-terrorism.
Experimental Study

Experimental study presentation explaining basic of RCTs, Types of experimental study and analysis including other essential topics.
CIGARETTE AND OTHER TOBACCO PRODUCTS ACT, (COTPA)

CIGARETTE AND OTHER TOBACCO PRODUCTS ACT AND VARIOUS SECTIONS
Review of current health service planning in Nepal from province to local level

Review of current health service planning in Nepal from province to local levelMohammad Aslam Shaiekh
Review of current health service planning in Nepal from province to the local level.ICD and ICF

ICD-10 is a statistical classification, which means that it contains a limited number of mutually exclusive code categories, which describe all disease concepts.
Pulse polio program and microplanning

The microplan is developed prior to the polio round by the Government and WHO-NPSP with support from partners at block, district and state levels. It gives a detailed plan of the house-to-house activity with team numbers, names of team members, supervisors, including influencers and routine immunization indicators.
Healthcare Delivery System in Federal Context of Nepal

Health care System in Federal Democratic Republic of Nepal
Cohort study and case control study

In brief about case control and cohort study used commonly in epidemiology.
National Leprosy Eradication Programme (NLEP)

National Leprosy Eradication Programme
National Leprosy Control and prevention programme
Risk factor surveillance of Non-communicable diseases

This presentation is based on the various W.H.O. available tools for risk factor surveillance of NCDS.
Health policy in developing countries

Decentralization
Tools of Policy making
Financing Health care
Public-Private Partnership
Health Research
International Organizations
Equity
Health Reforms in Developing Countries
Stake Holders
Primary health care

The course offers an opportunity to develop a holistic understanding of Primary Health Care, its functions, and scope. The course attendants will learn the principles of Primary Health Care, the course is expected to help the students to understand and internalize international health and public health transition facilitating the integration of health sector with other sectors.
More Related Content
What's hot
Public health emergencies

India being a developing country with growing population has been traditionally vulnerable to natural and man made disasters.
Development cannot be sustainable unless disaster mitigation is built into developmental process.
Disaster could be a nature calamity, outbreak of disease, bioterrorism, etc.
New Delhi, Feb 23. The health ministry has proposed a bill that seeks to empower state and local authorities to take appropriate actions to tackle public health emergencies like epidemics and bio-terrorism.
Experimental Study

Experimental study presentation explaining basic of RCTs, Types of experimental study and analysis including other essential topics.
CIGARETTE AND OTHER TOBACCO PRODUCTS ACT, (COTPA)

CIGARETTE AND OTHER TOBACCO PRODUCTS ACT AND VARIOUS SECTIONS
Review of current health service planning in Nepal from province to local level

Review of current health service planning in Nepal from province to local levelMohammad Aslam Shaiekh
Review of current health service planning in Nepal from province to the local level.ICD and ICF

ICD-10 is a statistical classification, which means that it contains a limited number of mutually exclusive code categories, which describe all disease concepts.
Pulse polio program and microplanning

The microplan is developed prior to the polio round by the Government and WHO-NPSP with support from partners at block, district and state levels. It gives a detailed plan of the house-to-house activity with team numbers, names of team members, supervisors, including influencers and routine immunization indicators.
Healthcare Delivery System in Federal Context of Nepal

Health care System in Federal Democratic Republic of Nepal
Cohort study and case control study

In brief about case control and cohort study used commonly in epidemiology.
National Leprosy Eradication Programme (NLEP)

National Leprosy Eradication Programme
National Leprosy Control and prevention programme
Risk factor surveillance of Non-communicable diseases

This presentation is based on the various W.H.O. available tools for risk factor surveillance of NCDS.
Health policy in developing countries

Decentralization
Tools of Policy making
Financing Health care
Public-Private Partnership
Health Research
International Organizations
Equity
Health Reforms in Developing Countries
Stake Holders
Primary health care

The course offers an opportunity to develop a holistic understanding of Primary Health Care, its functions, and scope. The course attendants will learn the principles of Primary Health Care, the course is expected to help the students to understand and internalize international health and public health transition facilitating the integration of health sector with other sectors.
What's hot (20)
Review of current health service planning in Nepal from province to local level

Review of current health service planning in Nepal from province to local level
Healthcare Delivery System in Federal Context of Nepal

Healthcare Delivery System in Federal Context of Nepal
Risk factor surveillance of Non-communicable diseases

Risk factor surveillance of Non-communicable diseases
Similar to Strobe checklist cross-sectional
Journal Club Prophylactic Intra-abdominal Drainage.pptx

Presentation with Journal Club about Prophylactic Intra-abdominal Drainage.pptx
A reporting guide for qualitative studies

1. How to report a qualitative research (i.e. how to draft a qualitative research paper)
Ich efficacy3 by Ramkrisna Bhunjawa 

It is about Efficacy Guidline of International Conference of Hormonization.
Literature Critique InstructionsStudents will complete and submit .docx

Literature Critique Instructions
Students will complete and submit a critique of an assigned literature. A modified version of the CONSORT (Consolidated Standards of Reporting Trials) 2010 checklist will be utilized to complete and grade this assignment. Total attainable points in this assignment is 45 points which accounts for 15% of the overall course grade. Students are highly encouraged to read “CONSORT 2010 Statement: Updated Guidelines for Reporting Parallel Group Randomized Trials.” and “CONSORT 2010 Explanation and Elaboration: updated guidelines for reporting parallel group randomized trials.” well in advance of the literature critique due date. These articles serve as a great resource aiding in the completion of this assignment. A link to these articles as well as other resources can be found below.
Resources:
· CONSORT website: http://www.consort-statement.org/home/
· A useful resource for general background information regarding CONSORT
· Provides examples of good reporting
· Offers resources glossary of terminology, links to published literature, and more
· CONSORT 2010 Statement: Updated Guidelines for Reporting Parallel Group Randomized Trials. http://annals.org/article.aspx?articleid=745807
· CONSORT 2010 Explanation and Elaboration: updated guidelines for reporting parallel group randomized trials.
http://www.consort-statement.org/Media/Default/Downloads/CONSORT%202010%20Explanation%20and%20Elaboration%20Document-BMJ.pdf
Course Number and Name
PHAR 610—Evidence Based Practice 2 Literature Critique & Journal Club
3
CONSORT 2010 checklist of information to include when reporting a randomised trial*
Section/Topic
Item No
Checklist item (each item is worth 1 point)
Reported on page No
& Line(s)
Title and abstract
1a
Identification as a randomised trial in the title
1b
Structured summary of trial design, methods, results, and conclusions (for specific guidance see CONSORT for abstracts)
Introduction
Background and objectives
2a
Scientific background and explanation of rationale
2b
Specific objectives or hypotheses
Methods
Trial design
3a
Description of trial design (such as parallel, factorial) including allocation ratio
3b
Important changes to methods after trial commencement (such as eligibility criteria), with reasons
Participants
4a
Eligibility criteria for participants
4b
Settings and locations where the data were collected
Interventions
5
The interventions for each group with sufficient details to allow replication, including how and when they were actually administered
Outcomes
6a
Completely defined pre-specified primary and secondary outcome measures, including how and when they were assessed
6b
Any changes to trial outcomes after the trial commenced, with reasons
Sample size
7a
How sample size was determined
7b
When applicable, explanation of any interim analyses and stopping guidelines
Randomisation:
Sequence
8a
Method used to generate the random allocation sequence
gener.
Literature Critique InstructionsStudents will complete and submit .docx

Literature Critique Instructions
Students will complete and submit a critique of an assigned literature. A modified version of the CONSORT (Consolidated Standards of Reporting Trials) 2010 checklist will be utilized to complete and grade this assignment. Total attainable points in this assignment is 45 points which accounts for 15% of the overall course grade. Students are highly encouraged to read “CONSORT 2010 Statement: Updated Guidelines for Reporting Parallel Group Randomized Trials.” and “CONSORT 2010 Explanation and Elaboration: updated guidelines for reporting parallel group randomized trials.” well in advance of the literature critique due date. These articles serve as a great resource aiding in the completion of this assignment. A link to these articles as well as other resources can be found below.
Resources:
· CONSORT website: http://www.consort-statement.org/home/
· A useful resource for general background information regarding CONSORT
· Provides examples of good reporting
· Offers resources glossary of terminology, links to published literature, and more
· CONSORT 2010 Statement: Updated Guidelines for Reporting Parallel Group Randomized Trials. http://annals.org/article.aspx?articleid=745807
· CONSORT 2010 Explanation and Elaboration: updated guidelines for reporting parallel group randomized trials.
http://www.consort-statement.org/Media/Default/Downloads/CONSORT%202010%20Explanation%20and%20Elaboration%20Document-BMJ.pdf
Course Number and Name
PHAR 610—Evidence Based Practice 2 Literature Critique & Journal Club
3
CONSORT 2010 checklist of information to include when reporting a randomised trial*
Section/Topic
Item No
Checklist item (each item is worth 1 point)
Reported on page No
& Line(s)
Title and abstract
1a
Identification as a randomised trial in the title
1b
Structured summary of trial design, methods, results, and conclusions (for specific guidance see CONSORT for abstracts)
Introduction
Background and objectives
2a
Scientific background and explanation of rationale
2b
Specific objectives or hypotheses
Methods
Trial design
3a
Description of trial design (such as parallel, factorial) including allocation ratio
3b
Important changes to methods after trial commencement (such as eligibility criteria), with reasons
Participants
4a
Eligibility criteria for participants
4b
Settings and locations where the data were collected
Interventions
5
The interventions for each group with sufficient details to allow replication, including how and when they were actually administered
Outcomes
6a
Completely defined pre-specified primary and secondary outcome measures, including how and when they were assessed
6b
Any changes to trial outcomes after the trial commenced, with reasons
Sample size
7a
How sample size was determined
7b
When applicable, explanation of any interim analyses and stopping guidelines
Randomisation:
Sequence
8a
Method used to generate the random allocation sequence
gener ...
Similar to Strobe checklist cross-sectional (20)
Journal Club Prophylactic Intra-abdominal Drainage.pptx

Journal Club Prophylactic Intra-abdominal Drainage.pptx
Literature Critique InstructionsStudents will complete and submit .docx

Literature Critique InstructionsStudents will complete and submit .docx
Literature Critique InstructionsStudents will complete and submit .docx

Literature Critique InstructionsStudents will complete and submit .docx
Recently uploaded
哪里卖(usq毕业证书)南昆士兰大学毕业证研究生文凭证书托福证书原版一模一样

原版定制【Q微信:741003700】《(usq毕业证书)南昆士兰大学毕业证研究生文凭证书》【Q微信:741003700】成绩单 、雅思、外壳、留信学历认证永久存档查询,采用学校原版纸张、特殊工艺完全按照原版一比一制作(包括:隐形水印,阴影底纹,钢印LOGO烫金烫银,LOGO烫金烫银复合重叠,文字图案浮雕,激光镭射,紫外荧光,温感,复印防伪)行业标杆!精益求精,诚心合作,真诚制作!多年品质 ,按需精细制作,24小时接单,全套进口原装设备,十五年致力于帮助留学生解决难题,业务范围有加拿大、英国、澳洲、韩国、美国、新加坡,新西兰等学历材料,包您满意。
【业务选择办理准则】
一、工作未确定,回国需先给父母、亲戚朋友看下文凭的情况,办理一份就读学校的毕业证【Q微信741003700】文凭即可
二、回国进私企、外企、自己做生意的情况,这些单位是不查询毕业证真伪的,而且国内没有渠道去查询国外文凭的真假,也不需要提供真实教育部认证。鉴于此,办理一份毕业证【微信741003700】即可
三、进国企,银行,事业单位,考公务员等等,这些单位是必需要提供真实教育部认证的,办理教育部认证所需资料众多且烦琐,所有材料您都必须提供原件,我们凭借丰富的经验,快捷的绿色通道帮您快速整合材料,让您少走弯路。
留信网认证的作用:
1:该专业认证可证明留学生真实身份
2:同时对留学生所学专业登记给予评定
3:国家专业人才认证中心颁发入库证书
4:这个认证书并且可以归档倒地方
5:凡事获得留信网入网的信息将会逐步更新到个人身份内,将在公安局网内查询个人身份证信息后,同步读取人才网入库信息
6:个人职称评审加20分
7:个人信誉贷款加10分
8:在国家人才网主办的国家网络招聘大会中纳入资料,供国家高端企业选择人才
【关于价格问题(保证一手价格)】
我们所定的价格是非常合理的,而且我们现在做得单子大多数都是代理和回头客户介绍的所以一般现在有新的单子 我给客户的都是第一手的代理价格,因为我想坦诚对待大家 不想跟大家在价格方面浪费时间
对于老客户或者被老客户介绍过来的朋友,我们都会适当给一些优惠。
一比一原版(Deakin毕业证书)迪肯大学毕业证如何办理

原版定制【微信:41543339】【(Deakin毕业证书)迪肯大学毕业证】【微信:41543339】成绩单、外壳、offer、留信学历认证(永久存档真实可查)采用学校原版纸张、特殊工艺完全按照原版一比一制作(包括:隐形水印,阴影底纹,钢印LOGO烫金烫银,LOGO烫金烫银复合重叠,文字图案浮雕,激光镭射,紫外荧光,温感,复印防伪)行业标杆!精益求精,诚心合作,真诚制作!多年品质 ,按需精细制作,24小时接单,全套进口原装设备,十五年致力于帮助留学生解决难题,业务范围有加拿大、英国、澳洲、韩国、美国、新加坡,新西兰等学历材料,包您满意。
【我们承诺采用的是学校原版纸张(纸质、底色、纹路),我们拥有全套进口原装设备,特殊工艺都是采用不同机器制作,仿真度基本可以达到100%,所有工艺效果都可提前给客户展示,不满意可以根据客户要求进行调整,直到满意为止!】
【业务选择办理准则】
一、工作未确定,回国需先给父母、亲戚朋友看下文凭的情况,办理一份就读学校的毕业证【微信41543339】文凭即可
二、回国进私企、外企、自己做生意的情况,这些单位是不查询毕业证真伪的,而且国内没有渠道去查询国外文凭的真假,也不需要提供真实教育部认证。鉴于此,办理一份毕业证【微信41543339】即可
三、进国企,银行,事业单位,考公务员等等,这些单位是必需要提供真实教育部认证的,办理教育部认证所需资料众多且烦琐,所有材料您都必须提供原件,我们凭借丰富的经验,快捷的绿色通道帮您快速整合材料,让您少走弯路。
留信网认证的作用:
1:该专业认证可证明留学生真实身份
2:同时对留学生所学专业登记给予评定
3:国家专业人才认证中心颁发入库证书
4:这个认证书并且可以归档倒地方
5:凡事获得留信网入网的信息将会逐步更新到个人身份内,将在公安局网内查询个人身份证信息后,同步读取人才网入库信息
6:个人职称评审加20分
7:个人信誉贷款加10分
8:在国家人才网主办的国家网络招聘大会中纳入资料,供国家高端企业选择人才
留信网服务项目:
1、留学生专业人才库服务(留信分析)
2、国(境)学习人员提供就业推荐信服务
3、留学人员区块链存储服务
→ 【关于价格问题(保证一手价格)】
我们所定的价格是非常合理的,而且我们现在做得单子大多数都是代理和回头客户介绍的所以一般现在有新的单子 我给客户的都是第一手的代理价格,因为我想坦诚对待大家 不想跟大家在价格方面浪费时间
对于老客户或者被老客户介绍过来的朋友,我们都会适当给一些优惠。
选择实体注册公司办理,更放心,更安全!我们的承诺:客户在留信官方认证查询网站查询到认证通过结果后付款,不成功不收费!
做(mqu毕业证书)麦考瑞大学毕业证硕士文凭证书学费发票原版一模一样

原版定制【Q微信:741003700】《(mqu毕业证书)麦考瑞大学毕业证硕士文凭证书》【Q微信:741003700】成绩单 、雅思、外壳、留信学历认证永久存档查询,采用学校原版纸张、特殊工艺完全按照原版一比一制作(包括:隐形水印,阴影底纹,钢印LOGO烫金烫银,LOGO烫金烫银复合重叠,文字图案浮雕,激光镭射,紫外荧光,温感,复印防伪)行业标杆!精益求精,诚心合作,真诚制作!多年品质 ,按需精细制作,24小时接单,全套进口原装设备,十五年致力于帮助留学生解决难题,业务范围有加拿大、英国、澳洲、韩国、美国、新加坡,新西兰等学历材料,包您满意。
【业务选择办理准则】
一、工作未确定,回国需先给父母、亲戚朋友看下文凭的情况,办理一份就读学校的毕业证【Q微信741003700】文凭即可
二、回国进私企、外企、自己做生意的情况,这些单位是不查询毕业证真伪的,而且国内没有渠道去查询国外文凭的真假,也不需要提供真实教育部认证。鉴于此,办理一份毕业证【微信741003700】即可
三、进国企,银行,事业单位,考公务员等等,这些单位是必需要提供真实教育部认证的,办理教育部认证所需资料众多且烦琐,所有材料您都必须提供原件,我们凭借丰富的经验,快捷的绿色通道帮您快速整合材料,让您少走弯路。
留信网认证的作用:
1:该专业认证可证明留学生真实身份
2:同时对留学生所学专业登记给予评定
3:国家专业人才认证中心颁发入库证书
4:这个认证书并且可以归档倒地方
5:凡事获得留信网入网的信息将会逐步更新到个人身份内,将在公安局网内查询个人身份证信息后,同步读取人才网入库信息
6:个人职称评审加20分
7:个人信誉贷款加10分
8:在国家人才网主办的国家网络招聘大会中纳入资料,供国家高端企业选择人才
【关于价格问题(保证一手价格)】
我们所定的价格是非常合理的,而且我们现在做得单子大多数都是代理和回头客户介绍的所以一般现在有新的单子 我给客户的都是第一手的代理价格,因为我想坦诚对待大家 不想跟大家在价格方面浪费时间
对于老客户或者被老客户介绍过来的朋友,我们都会适当给一些优惠。
Levelwise PageRank with Loop-Based Dead End Handling Strategy : SHORT REPORT ...

Abstract — Levelwise PageRank is an alternative method of PageRank computation which decomposes the input graph into a directed acyclic block-graph of strongly connected components, and processes them in topological order, one level at a time. This enables calculation for ranks in a distributed fashion without per-iteration communication, unlike the standard method where all vertices are processed in each iteration. It however comes with a precondition of the absence of dead ends in the input graph. Here, the native non-distributed performance of Levelwise PageRank was compared against Monolithic PageRank on a CPU as well as a GPU. To ensure a fair comparison, Monolithic PageRank was also performed on a graph where vertices were split by components. Results indicate that Levelwise PageRank is about as fast as Monolithic PageRank on the CPU, but quite a bit slower on the GPU. Slowdown on the GPU is likely caused by a large submission of small workloads, and expected to be non-issue when the computation is performed on massive graphs.
社内勉強会資料_LLM Agents .

単純な応答だけでなく、複雑な課題に対応できる柔軟性を持つLLM Agents。主要コンポーネントのPlanning、Memory、Tool Useについて解説しています。
一比一原版(UVic毕业证)维多利亚大学毕业证成绩单

UVic毕业证【微信95270640】(维多利亚大学毕业证成绩单本科学历)Q微信95270640(补办UVic学位文凭证书)维多利亚大学留信网学历认证怎么办理维多利亚大学毕业证成绩单精仿本科学位证书硕士文凭证书认证Seneca College diplomaoffer,Transcript办理硕士学位证书造假维多利亚大学假文凭学位证书制作UVic本科毕业证书硕士学位证书精仿维多利亚大学学历认证成绩单修改制作,办理真实认证、留信认证、使馆公证、购买成绩单,购买假文凭,购买假学位证,制造假国外大学文凭、毕业公证、毕业证明书、录取通知书、Offer、在读证明、雅思托福成绩单、假文凭、假毕业证、请假条、国际驾照、网上存档可查!
【实体公司】办维多利亚大学维多利亚大学毕业证文凭证书学历认证学位证文凭认证办留信网认证办留服认证办教育部认证(网上可查实体公司专业可靠)
— — — 留学归国服务中心 — — -
【主营项目】
一.维多利亚大学毕业证成绩单使馆认证教育部认证成绩单等!
二.真实使馆公证(即留学回国人员证明,不成功不收费)
三.真实教育部学历学位认证(教育部存档!教育部留服网站永久可查)
四.办理各国各大学文凭(一对一专业服务,可全程监控跟踪进度)
国外毕业证学位证成绩单办理流程:
1客户提供维多利亚大学维多利亚大学毕业证文凭证书办理信息:姓名生日专业学位毕业时间等(如信息不确定可以咨询顾问:我们有专业老师帮你查询);
2开始安排制作毕业证成绩单电子图;
3毕业证成绩单电子版做好以后发送给您确认;
4毕业证成绩单电子版您确认信息无误之后安排制作成品;
5成品做好拍照或者视频给您确认;
6快递给客户(国内顺丰国外DHLUPS等快读邮寄)。
专业服务请勿犹豫联系我!本公司是留学创业和海归创业者们的桥梁。一次办理终生受用一步到位高效服务。详情请在线咨询办理,欢迎有诚意办理的客户咨询!洽谈。
招聘代理:本公司诚聘英国加拿大澳洲新西兰美国法国德国新加坡各地代理人员如果你有业余时间有兴趣就请联系我们咨询顾问:+微信:95270640刀劈开抑或用拳头砸开每人抱起一大块就啃啃得满嘴满脸猴屁股般的红艳大家一个劲地指着对方吃吃地笑瓜裂得古怪奇形怪状却丝毫不影响瓜味甜丝丝的满嘴生津遍地都是瓜横七竖八的活像掷满了一地的大石块摘走二三只爷爷是断然发现不了的即便发现爷爷也不恼反而教山娃辨认孰熟孰嫩孰甜孰淡名义上是护瓜往往在瓜棚里坐上一刻饱吃一顿后山娃就领着阿黑漫山遍野地跑阿黑是一条黑色的大猎狗挺机灵的是山娃多年的忠实伙伴平时山娃上学阿黑也静
Adjusting primitives for graph : SHORT REPORT / NOTES

Graph algorithms, like PageRank Compressed Sparse Row (CSR) is an adjacency-list based graph representation that is
Multiply with different modes (map)
1. Performance of sequential execution based vs OpenMP based vector multiply.
2. Comparing various launch configs for CUDA based vector multiply.
Sum with different storage types (reduce)
1. Performance of vector element sum using float vs bfloat16 as the storage type.
Sum with different modes (reduce)
1. Performance of sequential execution based vs OpenMP based vector element sum.
2. Performance of memcpy vs in-place based CUDA based vector element sum.
3. Comparing various launch configs for CUDA based vector element sum (memcpy).
4. Comparing various launch configs for CUDA based vector element sum (in-place).
Sum with in-place strategies of CUDA mode (reduce)
1. Comparing various launch configs for CUDA based vector element sum (in-place).
The affect of service quality and online reviews on customer loyalty in the E...

Journal of Management and Creative Business.published book./
Q1’2024 Update: MYCI’s Leap Year Rebound

Key things you need to know on consumer confidence, key behavioral, tech, e-wallet & esports trends in Malaysia.
一比一原版(CBU毕业证)不列颠海角大学毕业证成绩单

CBU毕业证【微信95270640】《如何办理不列颠海角大学毕业证认证》【办证Q微信95270640】《不列颠海角大学文凭毕业证制作》《CBU学历学位证书哪里买》办理不列颠海角大学学位证书扫描件、办理不列颠海角大学雅思证书!
国际留学归国服务中心《如何办不列颠海角大学毕业证认证》《CBU学位证书扫描件哪里买》实体公司,注册经营,行业标杆,精益求精!
1:1完美还原海外各大学毕业材料上的工艺:水印阴影底纹钢印LOGO烫金烫银LOGO烫金烫银复合重叠。文字图案浮雕激光镭射紫外荧光温感复印防伪。
可办理以下真实不列颠海角大学存档留学生信息存档认证:
1不列颠海角大学真实留信网认证(网上可查永久存档无风险百分百成功入库);
2真实教育部认证(留服)等一切高仿或者真实可查认证服务(暂时不可办理);
3购买英美真实学籍(不用正常就读直接出学历);
4英美一年硕士保毕业证项目(保录取学校挂名不用正常就读保毕业)
留学本科/硕士毕业证书成绩单制作流程:
1客户提供办理信息:姓名生日专业学位毕业时间等(如信息不确定可以咨询顾问:我们有专业老师帮你查询不列颠海角大学不列颠海角大学本科学位证成绩单);
2开始安排制作不列颠海角大学毕业证成绩单电子图;
3不列颠海角大学毕业证成绩单电子版做好以后发送给您确认;
4不列颠海角大学毕业证成绩单电子版您确认信息无误之后安排制作成品;
5不列颠海角大学成品做好拍照或者视频给您确认;
6快递给客户(国内顺丰国外DHLUPS等快读邮寄)
— — — — — — — — — — — 《文凭顾问Q/微:95270640》这么大这么美的地方赚大钱高楼大厦鳞次栉比大街小巷人潮涌动山娃一路张望一路惊叹他发现城里的桥居然层层叠叠扭来扭去桥下没水却有着水一般的车水马龙山娃惊诧于城里的公交车那么大那么美不用买票乖乖地掷下二枚硬币空调享受还能坐着看电视呢屡经辗转山娃终于跟着父亲到家了山娃没想到父亲城里的家会如此寒碜更没料到父亲的城里竟有如此简陋的鬼地方父亲的家在高楼最底屋最下面很矮很黑是很不显眼的地下室父亲的家安在别人脚底下孰
Empowering Data Analytics Ecosystem.pptx

Show drafts
volume_up
Empowering the Data Analytics Ecosystem: A Laser Focus on Value
The data analytics ecosystem thrives when every component functions at its peak, unlocking the true potential of data. Here's a laser focus on key areas for an empowered ecosystem:
1. Democratize Access, Not Data:
Granular Access Controls: Provide users with self-service tools tailored to their specific needs, preventing data overload and misuse.
Data Catalogs: Implement robust data catalogs for easy discovery and understanding of available data sources.
2. Foster Collaboration with Clear Roles:
Data Mesh Architecture: Break down data silos by creating a distributed data ownership model with clear ownership and responsibilities.
Collaborative Workspaces: Utilize interactive platforms where data scientists, analysts, and domain experts can work seamlessly together.
3. Leverage Advanced Analytics Strategically:
AI-powered Automation: Automate repetitive tasks like data cleaning and feature engineering, freeing up data talent for higher-level analysis.
Right-Tool Selection: Strategically choose the most effective advanced analytics techniques (e.g., AI, ML) based on specific business problems.
4. Prioritize Data Quality with Automation:
Automated Data Validation: Implement automated data quality checks to identify and rectify errors at the source, minimizing downstream issues.
Data Lineage Tracking: Track the flow of data throughout the ecosystem, ensuring transparency and facilitating root cause analysis for errors.
5. Cultivate a Data-Driven Mindset:
Metrics-Driven Performance Management: Align KPIs and performance metrics with data-driven insights to ensure actionable decision making.
Data Storytelling Workshops: Equip stakeholders with the skills to translate complex data findings into compelling narratives that drive action.
Benefits of a Precise Ecosystem:
Sharpened Focus: Precise access and clear roles ensure everyone works with the most relevant data, maximizing efficiency.
Actionable Insights: Strategic analytics and automated quality checks lead to more reliable and actionable data insights.
Continuous Improvement: Data-driven performance management fosters a culture of learning and continuous improvement.
Sustainable Growth: Empowered by data, organizations can make informed decisions to drive sustainable growth and innovation.
By focusing on these precise actions, organizations can create an empowered data analytics ecosystem that delivers real value by driving data-driven decisions and maximizing the return on their data investment.
一比一原版(QU毕业证)皇后大学毕业证成绩单

QU毕业证【微信95270640】办理皇后大学毕业证原版一模一样、QU毕业证制作【Q微信95270640】《皇后大学毕业证购买流程》《QU成绩单制作》皇后大学毕业证书QU毕业证文凭皇后大学
本科毕业证书,学历学位认证如何办理【留学国外学位学历认证、毕业证、成绩单、大学Offer、雅思托福代考、语言证书、学生卡、高仿教育部认证等一切高仿或者真实可查认证服务】代办国外(海外)英国、加拿大、美国、新西兰、澳大利亚、新西兰等国外各大学毕业证、文凭学历证书、成绩单、学历学位认证真实可查。
1:1完美还原海外各大学毕业材料上的工艺:水印阴影底纹钢印LOGO烫金烫银LOGO烫金烫银复合重叠。文字图案浮雕激光镭射紫外荧光温感复印防伪。
可办理以下真实皇后大学存档留学生信息存档认证:
1皇后大学真实留信网认证(网上可查永久存档无风险百分百成功入库);
2真实教育部认证(留服)等一切高仿或者真实可查认证服务(暂时不可办理);
3购买英美真实学籍(不用正常就读直接出学历);
4英美一年硕士保毕业证项目(保录取学校挂名不用正常就读保毕业)
留学本科/硕士毕业证书成绩单制作流程:
1客户提供办理信息:姓名生日专业学位毕业时间等(如信息不确定可以咨询顾问:我们有专业老师帮你查询皇后大学皇后大学硕士毕业证成绩单);
2开始安排制作皇后大学毕业证成绩单电子图;
3皇后大学毕业证成绩单电子版做好以后发送给您确认;
4皇后大学毕业证成绩单电子版您确认信息无误之后安排制作成品;
5皇后大学成品做好拍照或者视频给您确认;
6快递给客户(国内顺丰国外DHLUPS等快读邮寄)
— — — — — — — — — — — 《文凭顾问Q/微:95270640》很感动很无奈房东的儿子小伍笨手笨脚的不会说普通话满口粤语态度十分傲慢一副盛气凌人的样子山娃试图接近他跟他交友与城里人交友但他俩就好像是两个世界里的人根本拢不到一块儿不知不觉山娃倒跟周围出租屋里的几个小伙伴成了好朋友因为他们也是从乡下进城过暑假的小学生快乐的日子总是过得飞快山娃尚未完全认清那几位小朋友时他们却一个接一个地回家了山娃这时才恍然发现二个月的暑假已转到了尽头他的城市生活也将划上一个不很圆义
Data Centers - Striving Within A Narrow Range - Research Report - MCG - May 2...

M Capital Group (“MCG”) expects to see demand and the changing evolution of supply, facilitated through institutional investment rotation out of offices and into work from home (“WFH”), while the ever-expanding need for data storage as global internet usage expands, with experts predicting 5.3 billion users by 2023. These market factors will be underpinned by technological changes, such as progressing cloud services and edge sites, allowing the industry to see strong expected annual growth of 13% over the next 4 years.
Whilst competitive headwinds remain, represented through the recent second bankruptcy filing of Sungard, which blames “COVID-19 and other macroeconomic trends including delayed customer spending decisions, insourcing and reductions in IT spending, energy inflation and reduction in demand for certain services”, the industry has seen key adjustments, where MCG believes that engineering cost management and technological innovation will be paramount to success.
MCG reports that the more favorable market conditions expected over the next few years, helped by the winding down of pandemic restrictions and a hybrid working environment will be driving market momentum forward. The continuous injection of capital by alternative investment firms, as well as the growing infrastructural investment from cloud service providers and social media companies, whose revenues are expected to grow over 3.6x larger by value in 2026, will likely help propel center provision and innovation. These factors paint a promising picture for the industry players that offset rising input costs and adapt to new technologies.
According to M Capital Group: “Specifically, the long-term cost-saving opportunities available from the rise of remote managing will likely aid value growth for the industry. Through margin optimization and further availability of capital for reinvestment, strong players will maintain their competitive foothold, while weaker players exit the market to balance supply and demand.”
一比一原版(BU毕业证)波士顿大学毕业证成绩单

BU毕业证【微信95270640】购买(波士顿大学毕业证成绩单硕士学历)Q微信95270640代办BU学历认证留信网伪造波士顿大学学位证书精仿波士顿大学本科/硕士文凭证书补办波士顿大学 diplomaoffer,Transcript购买波士顿大学毕业证成绩单购买BU假毕业证学位证书购买伪造波士顿大学文凭证书学位证书,专业办理雅思、托福成绩单,学生ID卡,在读证明,海外各大学offer录取通知书,毕业证书,成绩单,文凭等材料:1:1完美还原毕业证、offer录取通知书、学生卡等各种在读或毕业材料的防伪工艺(包括 烫金、烫银、钢印、底纹、凹凸版、水印、防伪光标、热敏防伪、文字图案浮雕,激光镭射,紫外荧光,温感光标)学校原版上有的工艺我们一样不会少,不论是老版本还是最新版本,都能保证最高程度还原,力争完美以求让所有同学都能享受到完美的品质服务。
专业为留学生办理波士顿大学波士顿大学毕业证offer【100%存档可查】留学全套申请材料办理。本公司承诺所有毕业证成绩单成品全部按照学校原版工艺对照一比一制作和学校一样的羊皮纸张保证您证书的质量!
如果你回国在学历认证方面有以下难题请联系我们我们将竭诚为你解决认证瓶颈
1所有材料真实但资料不全无法提供完全齐整的原件。【如:成绩单丶毕业证丶回国证明等材料中有遗失的。】
2获得真实的国外最终学历学位但国外本科学历就读经历存在问题或缺陷。【如:国外本科是教育部不承认的或者是联合办学项目教育部没有备案的或者外本科没有正常毕业的。】
3学分转移联合办学等情况复杂不知道怎么整理材料的。时间紧迫自己不清楚递交流程的。
如果你是以上情况之一请联系我们我们将在第一时间内给你免费咨询相关信息。我们将帮助你整理认证所需的各种材料.帮你解决国外学历认证难题。
国外波士顿大学波士顿大学毕业证offer办理方法:
1客户提供办理信息:姓名生日专业学位毕业时间等(如信息不确定可以咨询顾问:我们有专业老师帮你查询波士顿大学波士顿大学毕业证offer);
2开始安排制作波士顿大学毕业证成绩单电子图;
3波士顿大学毕业证成绩单电子版做好以后发送给您确认;
4波士顿大学毕业证成绩单电子版您确认信息无误之后安排制作成品;
5波士顿大学成品做好拍照或者视频给您确认;
6快递给客户(国内顺丰国外DHLUPS等快读邮寄)。二条巴掌般大的裤衩衩走出泳池山娃感觉透身粘粘乎乎散发着药水味有点痒山娃顿时留恋起家乡的小河潺潺活水清凉无比日子就这样孤寂而快乐地过着寂寞之余山娃最神往最开心就是晚上无论多晚多累父亲总要携山娃出去兜风逛夜市流光溢彩人潮涌动的都市夜生活总让山娃目不暇接惊叹不已父亲老问山娃想买什么想吃什么山娃知道父亲赚钱很辛苦除了书籍和文具山娃啥也不要能牵着父亲的手满城闲逛他已心满意足了父亲连挑了三套童装叫山娃试穿山伸
Ch03-Managing the Object-Oriented Information Systems Project a.pdf

Object oriented system analysis and design
一比一原版(UofS毕业证书)萨省大学毕业证如何办理

原版定制【微信:41543339】【(UofS毕业证书)萨省大学毕业证】【微信:41543339】成绩单、外壳、offer、留信学历认证(永久存档真实可查)采用学校原版纸张、特殊工艺完全按照原版一比一制作(包括:隐形水印,阴影底纹,钢印LOGO烫金烫银,LOGO烫金烫银复合重叠,文字图案浮雕,激光镭射,紫外荧光,温感,复印防伪)行业标杆!精益求精,诚心合作,真诚制作!多年品质 ,按需精细制作,24小时接单,全套进口原装设备,十五年致力于帮助留学生解决难题,业务范围有加拿大、英国、澳洲、韩国、美国、新加坡,新西兰等学历材料,包您满意。
【我们承诺采用的是学校原版纸张(纸质、底色、纹路),我们拥有全套进口原装设备,特殊工艺都是采用不同机器制作,仿真度基本可以达到100%,所有工艺效果都可提前给客户展示,不满意可以根据客户要求进行调整,直到满意为止!】
【业务选择办理准则】
一、工作未确定,回国需先给父母、亲戚朋友看下文凭的情况,办理一份就读学校的毕业证【微信41543339】文凭即可
二、回国进私企、外企、自己做生意的情况,这些单位是不查询毕业证真伪的,而且国内没有渠道去查询国外文凭的真假,也不需要提供真实教育部认证。鉴于此,办理一份毕业证【微信41543339】即可
三、进国企,银行,事业单位,考公务员等等,这些单位是必需要提供真实教育部认证的,办理教育部认证所需资料众多且烦琐,所有材料您都必须提供原件,我们凭借丰富的经验,快捷的绿色通道帮您快速整合材料,让您少走弯路。
留信网认证的作用:
1:该专业认证可证明留学生真实身份
2:同时对留学生所学专业登记给予评定
3:国家专业人才认证中心颁发入库证书
4:这个认证书并且可以归档倒地方
5:凡事获得留信网入网的信息将会逐步更新到个人身份内,将在公安局网内查询个人身份证信息后,同步读取人才网入库信息
6:个人职称评审加20分
7:个人信誉贷款加10分
8:在国家人才网主办的国家网络招聘大会中纳入资料,供国家高端企业选择人才
留信网服务项目:
1、留学生专业人才库服务(留信分析)
2、国(境)学习人员提供就业推荐信服务
3、留学人员区块链存储服务
→ 【关于价格问题(保证一手价格)】
我们所定的价格是非常合理的,而且我们现在做得单子大多数都是代理和回头客户介绍的所以一般现在有新的单子 我给客户的都是第一手的代理价格,因为我想坦诚对待大家 不想跟大家在价格方面浪费时间
对于老客户或者被老客户介绍过来的朋友,我们都会适当给一些优惠。
选择实体注册公司办理,更放心,更安全!我们的承诺:客户在留信官方认证查询网站查询到认证通过结果后付款,不成功不收费!
Recently uploaded (20)
Levelwise PageRank with Loop-Based Dead End Handling Strategy : SHORT REPORT ...

Levelwise PageRank with Loop-Based Dead End Handling Strategy : SHORT REPORT ...
Adjusting primitives for graph : SHORT REPORT / NOTES

Adjusting primitives for graph : SHORT REPORT / NOTES
The affect of service quality and online reviews on customer loyalty in the E...

The affect of service quality and online reviews on customer loyalty in the E...
Sample_Global Non-invasive Prenatal Testing (NIPT) Market, 2019-2030.pdf

Sample_Global Non-invasive Prenatal Testing (NIPT) Market, 2019-2030.pdf
Data Centers - Striving Within A Narrow Range - Research Report - MCG - May 2...

Data Centers - Striving Within A Narrow Range - Research Report - MCG - May 2...
Ch03-Managing the Object-Oriented Information Systems Project a.pdf

Ch03-Managing the Object-Oriented Information Systems Project a.pdf
Strobe checklist cross-sectional
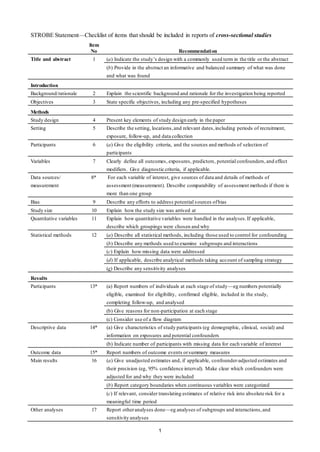
- 1. 1 STROBE Statement—Checklist of items that should be included in reports of cross-sectional studies Item No Recommendation Title and abstract 1 (a) Indicate the study’s design with a commonly used term in the title or the abstract (b) Provide in the abstract an informative and balanced summary of what was done and what was found Introduction Background/rationale 2 Explain the scientific background and rationale for the investigation being reported Objectives 3 State specific objectives, including any pre-specified hypotheses Methods Study design 4 Present key elements of study design early in the paper Setting 5 Describe the setting, locations,and relevant dates,including periods of recruitment, exposure, follow-up, and data collection Participants 6 (a) Give the eligibility criteria, and the sources and methods of selection of participants Variables 7 Clearly define all outcomes, exposures, predictors, potential confounders,and effect modifiers. Give diagnostic criteria, if applicable. Data sources/ measurement 8* For each variable of interest, give sources of data and details of methods of assessment (measurement). Describe comparability of assessment methods if there is more than one group Bias 9 Describe any efforts to address potential sources ofbias Study size 10 Explain how the study size was arrived at Quantitative variables 11 Explain how quantitative variables were handled in the analyses.If applicable, describe which groupings were chosen and why Statistical methods 12 (a) Describe all statistical methods, including those used to control for confounding (b) Describe any methods used to examine subgroups and interactions (c) Explain how missing data were addressed (d) If applicable, describe analytical methods taking account of sampling strategy (e) Describe any sensitivity analyses Results Participants 13* (a) Report numbers of individuals at each stage of study—eg numbers potentially eligible, examined for eligibility, confirmed eligible, included in the study, completing follow-up, and analysed (b) Give reasons for non-participation at each stage (c) Consider use of a flow diagram Descriptive data 14* (a) Give characteristics of study participants (eg demographic, clinical, social) and information on exposures and potential confounders (b) Indicate number of participants with missing data for each variable of interest Outcome data 15* Report numbers of outcome events orsummary measures Main results 16 (a) Give unadjusted estimates and, if applicable, confounder-adjusted estimates and their precision (eg, 95% confidence interval). Make clear which confounders were adjusted for and why they were included (b) Report category boundaries when continuous variables were categorized (c) If relevant, consider translating estimates of relative risk into absolute risk for a meaningful time period Other analyses 17 Report otheranalyses done—eg analyses of subgroups and interactions,and sensitivity analyses
- 2. 2 Discussion Key results 18 Summarise key results with reference to study objectives Limitations 19 Discuss limitations of the study,taking into account sources ofpotential bias or imprecision. Discuss both direction and magnitude of any potential bias Interpretation 20 Give a cautious overall interpretation of results considering objectives, limitations, multiplicity of analyses,results from similar studies,and other relevant evidence Generalisability 21 Discuss the generalisability (external validity) of the study results Other information Funding 22 Give the source of funding and the role of the funders for the present study and,if applicable, for the original study on which the present article is based *Give information separately for exposed and unexposed groups. Note: An Explanation and Elaboration article discusses each checklist item and gives methodological background and published examples of transparent reporting. The STROBE checklist is best used in conjunction with this article (freely available on the Web sites of PLoS Medicine at http://www.plosmedicine.org/, Annals of Internal Medicine at http://www.annals.org/, and Epidemiology at http://www.epidem.com/). Information on the STROBE Initiative is available at www.strobe-statement.org.
