The document discusses the simulation of the formaldehyde production process using a silver catalyst, conducted through Aspen HYSYS software. It provides an overview of formaldehyde's significance in various industries, details the simulation methodology, and examines key parameters such as temperature and pressure profiles, resulting in a product yield of 74%. The simulation helps visualize the production environment and aids in understanding the material and energy requirements for establishing a formaldehyde plant.
![1
Simulation of Formaldehyde Production Process
Ruhul Amin, Nazibul Islam, Rezwanul Islam, Yusuf Imtiaz, Saeed M., Unaiza M.
Department of Chemical Engineering
Bangladesh University of Engineering and Technology, Dhaka-1000, Bangladesh
Abstract
Formaldehyde plays a major role in the synthesis of many important compounds. Worldwide production
of formaldehyde is growing day bay day. There are various industrial processes for the production of
formaldehyde. This article starts with an overview of formaldehyde and the history of formaldehyde
production. Subsequently, production of formaldehyde using silver catalyst is simulated with the help of
Aspen Hysys 7.1. Important parameters such as temperature profile, pressure profile, fluid properties etc
were investigated with this simulation process. The effect of temperature in the reaction was also
examined. The simulation process validated that for maximum conversion to take place, the reaction must
occur in 550
C. Finally, 74% formaldehyde was obtained as product.
Key Words: Formaldehyde, Oxidation-dehydrogenation, NRTL, Simulation
1. Introduction
Formaldehyde was discovered in 1859 by a
Russian chemist named Aleksandr Butlerov.
However it was in 1869, that German chemist
August Hofmann developed a practical method
to synthesis formaldehyde from methanol. [1]
It
is a colorless gas with a distinctive pungent
order. It is highly flammable with a flashpoint of
500
C; the heat of combustion is 134.1kcal/mol
or 4.47kcal/g.[2]
Formaldehyde is soluble in a
variety of solvents and is miscible in water. [2, 3]
Formaldehyde is a key chemical component in
many manufacturing processes. It is used as a
building block for the synthesis of more
complex compounds and materials. [4]
In
approximate order of decreasing consumption,
products generated from formaldehyde include
urea formaldehyde resin, melamine resin, phenol
formaldehyde resin,poly-oxy-methylene plastics,
1,4-butane-di-ol, and methylene-di-phenyl-di-
iso-cyanate.[5]
In biomedical industry,
formaldehyde is used in vaccines, medicines,
plastics and in x-ray machines. The phenolic
molding resins produced from formaldehyde are
used in appliances, electrical control, telephone
and wiring devices. [6]
In the automotive and
building industries, formaldehyde-based acetal
resins are used in the electrical system,
transmission, engine block, door panels and
break shoes. [7]](https://image.slidesharecdn.com/1405099379-160311030409/75/Simulation-of-Formaldehyde-Production-Process-1-2048.jpg)
![2
The total annual formaldehyde capacity in 1998
was estimated by 11.3 billion pounds. Since then
and the production capacity around the globe is
expanding exponentially reaching a world’s
production of 32.5 million metric tons by 2012.
[7, 8]
There are two main routes for formaldehyde
production: oxidation-dehydrogenation using a
silver catalyst involving both the complete or
incomplete conversion of methanol; and the
direct oxidation of methanol to formaldehyde
using metal oxide catalysts. [9, 10]
In the
oxidation-dehydrogenation route, vaporized
methanol with air is passed over a thin bed of
silver-crystal catalyst at about 6500
C.
Formaldehyde is formed by the de-
hydrogenation of methanol. [11, 12]
The other
route involves the oxidation of methanol over a
catalyst of molybdenum and iron at 3500
C. [13]
This article deals with the detailed study of the
simulation of formaldehyde production from
methanol. Simulation has been done with the
help of Aspen Hysys v7.2. Although simulation
does not give the real world performance or the
real life production environment but if the basic
process is known and related data are available,
it is the best way by which an individual can get
ideas of an industrial process without conducting
any experiment.
2. Methodology
The process of producing formaldehyde from
methanol is simulated in Simulation software
Aspen Hyssy 7.1. Aspen Hysys is a simulation-
software which comes along with excellent
reference & tutorial manuals for simulating a
process. Hysys does not wait until entering
every process condition before beginning
calculation. It calculates as much as it can at all
time and results are always available, even
during calculation. Any changes that one makes
to the data are automatically propagated
throughout the program to anywhere that entry
appears and all necessary recalculations are
instantly carried out. It tends to be a lot easier to
catch errors as one gradually converge the
process simulation.
The Fluid package used in this simulation is
NRTL. The non-random two-liquid model is
known as NRTL equation in short[14]. NRTL is
an activity coefficient model that correlates
the activity coefficients of a compound i with
its mole fractions in the liquid phase concerned.
The concept of NRTL is based on the hypothesis
of Wilson that the local concentration around a
molecule is different from the bulk
concentration. This difference is due to a
difference between the interaction energy of the
central molecule with the molecules of its own
kind Uii and that with the molecules of the other
kind Uij. The energy difference also introduces a
non-randomness at the local molecular level.
The NRTL model belongs to the so-called local-
composition models. Other models of this type
are the Wilson model, the UNIQUAC model,
and the group contribution model UNIFAC.
These local-composition models are not](https://image.slidesharecdn.com/1405099379-160311030409/85/Simulation-of-Formaldehyde-Production-Process-2-320.jpg)
![3
thermodynamically consistent due to the
assumption that the local composition around
molecule i is independent of the local
composition around molecule j. This assumption
is not true, as was shown by Flemmer in
1976[15, 16].
2.1 Process Description
Formaldehyde results from the exothermic
oxidation and endothermic hydrogenation of
methanol. These two reactions occur
simultaneously in commercial units in a
balanced reaction, called auto thermal because
the oxidative reaction furnishes the heat to cause
the dehydrogenation to take place. About 50 to
60 percent of the formaldehyde is formed by the
exothermic reaction. The oxidation requires
1.6m3
of air per kilogram of methanol reacted, a
ratio that is maintained when passing separate
streams of these two materials forward. Fresh &
recycled methanol are vaporized, superheated
and passed into the methanol-air mixer.
Atmospheric air is purified, compressed and pre-
heated to 540
C in a finned heat exchanger. The
products leave the converter at 620o
C and at 34
to 69 KPa absolute. The converter is a small
water-jacketed vessel containing the silver-
catalyst. About 65 percent of the methanol is
converted per pass. The reactor effluent contains
about 25% formaldehyde, which is absorbed
with the excess methanol and piped to the make
tank. The latter feeds the methanol column for
separation of recycle methanol overhead, the
bottom stream containing the formaldehyde and
a few percent methanol. The water intake adjusts
the formaldehyde to 37% strength (marketed as
formalin). The yield from the reaction is 85 to
90 percent. The catalyst is easily poisoned so
stainless-steel equipment must be used to protect
the catalyst from metal contamination.
2.2 Simplified Block Diagram
Figure 1: Block diagram of the total process](https://image.slidesharecdn.com/1405099379-160311030409/85/Simulation-of-Formaldehyde-Production-Process-3-320.jpg)
![4
2.3 Set Stoichiometryand Rate of
Reaction
As mentioned earlier, Formaldehyde results
from the exothermic oxidation and endothermic
de-hydrogenation of methanol.
CH3OH + 1/2O2 CH2O + H2O; H = -156 KJ
CH3OH CH2O + H2; H = +85 KJ
So the stoichimetry for Methanol in the 1st
and
2nd
reaction would be -1
For Formaldehyde it would be +1 for the both
reactions.
For Oxygen it would be -0.5 in the 1st
reaction
And for water and Hydrogen it would be +1 for
the 1st
and 2nd
reaction respectively.
The rate of reaction for the first reaction will be:
−rm1[
mole
gcatalyst
hr
] =
k1pm
1 + k2pm
Where,
𝑙𝑛𝑘1 = 12.50 −
8774
T
And
𝑙𝑛𝑘2 = −17.29 +
7439
T
The rate of reaction for the second reaction will
be:
𝑟𝑚2[
𝑚𝑜𝑙𝑒
𝑔𝑐𝑎𝑡𝑎𝑙𝑦𝑠𝑡
ℎ𝑟
] =
K1√pm
1 + K2√pm
Where,
𝑙𝑛𝐾1 = 16.9 −
12500
T
And
𝑙𝑛𝐾2 = 25.0 −
15724
T
For all the equations, T is in Kelvin. [8]
2.4 The Simulation Environment
1. First Methanol and feed air are delivered
to a mixer and later preheated to 55⁰ C
and delivered to the reactor.
2. In the first step of the reaction, methanol
reacts with oxygen to give formaldehyde
and water
3. In the 2nd
stage some of the methanol
breaks up to formaldehyde and
hydrogen.
4. The vapor from the reactor outlet is
cooled to 10⁰ C and delivered to a
separator.
5. The Hydrogen is separated from the
mixture.
6. The remaining mixture is heated to 100⁰
C and fed to the distillation column.
7. From the distillation column, we get the
liquid product of 83.2% formaldehyde
and a vapor product of 44.1%
formaldehyde.
8. The vapor product is heated to 35⁰ C
and delivered to a storage tank.](https://image.slidesharecdn.com/1405099379-160311030409/85/Simulation-of-Formaldehyde-Production-Process-4-320.jpg)
![5
2.5 Importance of Temperature in
the Simulation
The heated feed that is delivered to the
first reactor is heated to a temperature of
55° C by delivering the feed to a heater.
It is heated to 55° C because this is the
optimum temperature of reactor inlet.
The final top product from the
distillation column is delivered to a
heater before it is stored in the storage
tank. The outlet from the heater is
heated to a temperature between 35-45°
C. At temperatures below 35° C, the
product forms formaldehyde polymer
which is not desired. Storage at
temperatures between 35-45° C further
inhibits the formation of formaldehyde
polymers[2]
.
3. Results and Discussions
Final composition of Formaldehyde obtained is
74.8%.
Different parameters of the distillation
column are shown in different graphs below-
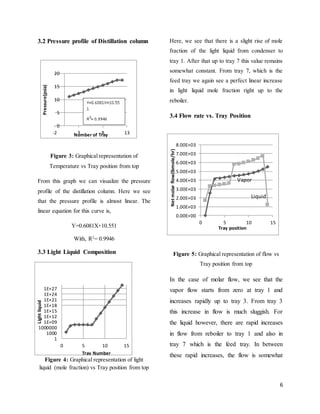
3.1 Temperature Profile of Distillation
Column
Figure 2: Graphical representation of
Temperature vs Tray position from top
From figure 2 we can see that the condenser
temperature is around -2500C. The
temperature rises rapidly from the condenser
and reaches near 1000C at stage 2 that is first
tray after the condenser. From tray 2, the
temperature rise is linear and is around
1100C in the reboiler.
-300
-200
-100
0
100
200
0 5 10 15
Temperature
Number of Tray](https://image.slidesharecdn.com/1405099379-160311030409/85/Simulation-of-Formaldehyde-Production-Process-5-320.jpg)