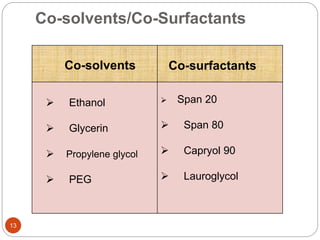
This document discusses Self-Emulsifying Drug Delivery Systems (SEDDS), which are isotropic mixtures of oils, surfactants, and co-solvents that can solubilize drugs and promote self-emulsification. SEDDS enhance oral drug bioavailability, protect drugs from the hostile gastrointestinal environment, and reduce variability. The document describes the components of SEDDS including oils, surfactants, co-surfactants and drugs. It also outlines the formulation process and methods to evaluate parameters like stability, dispersibility, droplet size and drug release. SEDDS are a promising approach for improving oral delivery of poorly soluble drugs.