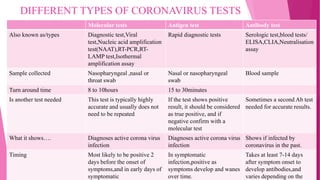
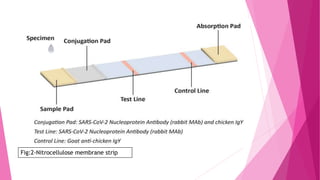
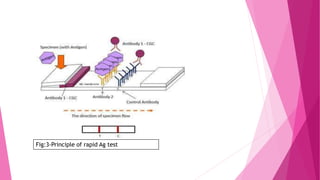
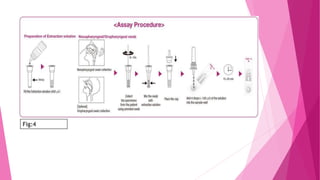
The document outlines the emergence and characteristics of the SARS-CoV-2 virus, which causes COVID-19, and details the rapid antigen test used for its detection. It explains the test's methodology, sample collection, and interpretation of results, emphasizing its importance in diagnosing COVID-19 quickly. Furthermore, it addresses safety precautions, limitations of the test, and references for further reading.