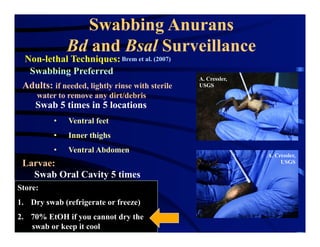
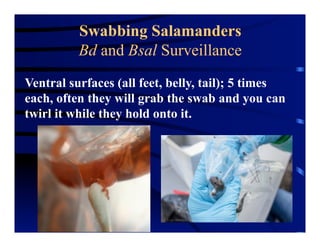
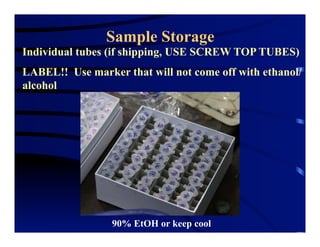
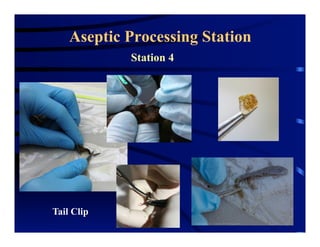
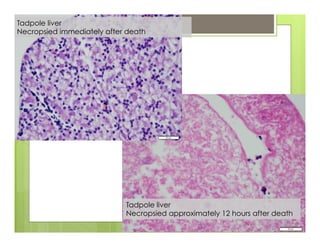
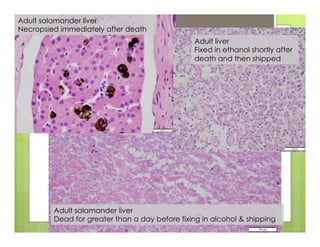
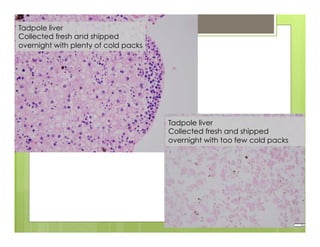
The document outlines procedures for the collection, processing, and shipping of amphibian samples for diagnostic testing, emphasizing biosecurity measures and proper sampling techniques. It discusses cohabitation experiments, swabbing methods, storage requirements, and the importance of using appropriate materials for shipping specimens. Additionally, it highlights the significance of timely specimen processing to avoid degradation and contamination that could affect diagnostic results.



![Co-housing Animals
Mean =
0.8 – 0.9 contacts/min for 40 tadpoles/m2
(10 tadpoles per 5-gal [19 L] bucket)
All should contact each other in 9 Minutes
10, 20, 40%
X
15, 30, 60 min
What about
gloves?
Ranavirus Example](https://image.slidesharecdn.com/miller-150825150909-lva1-app6891/85/Sample-collection-and-shipping-what-s-important-and-why-4-320.jpg)