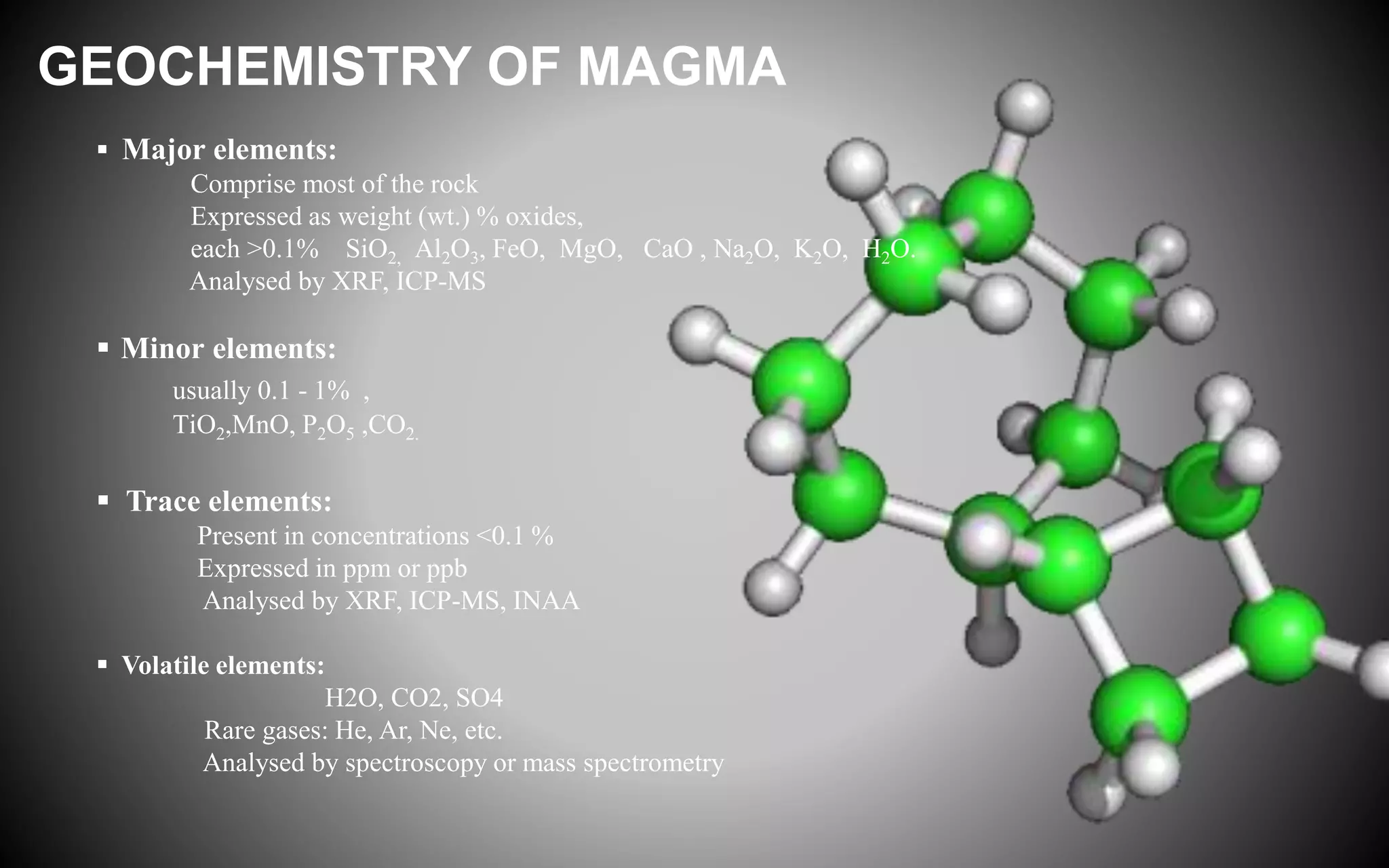
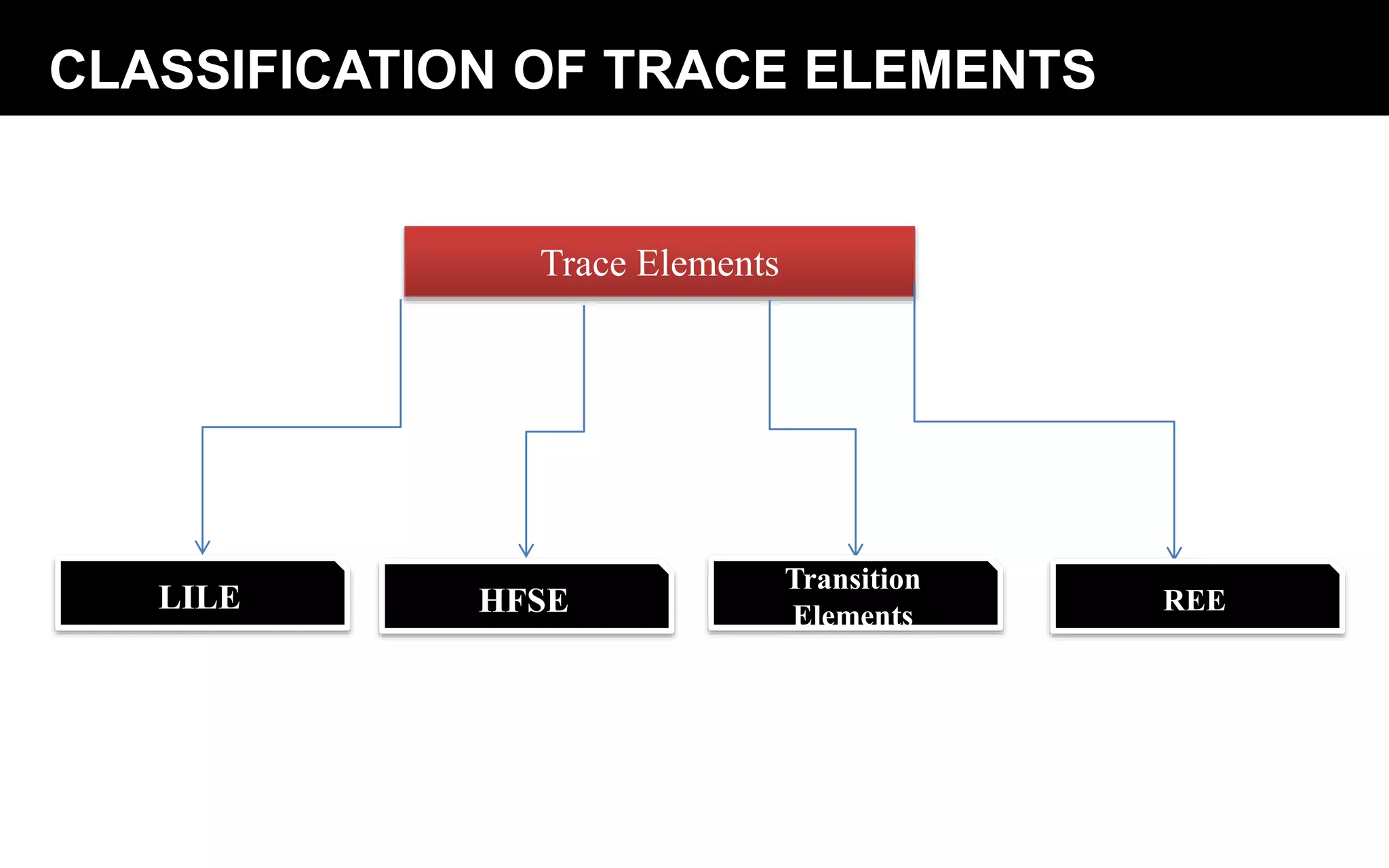
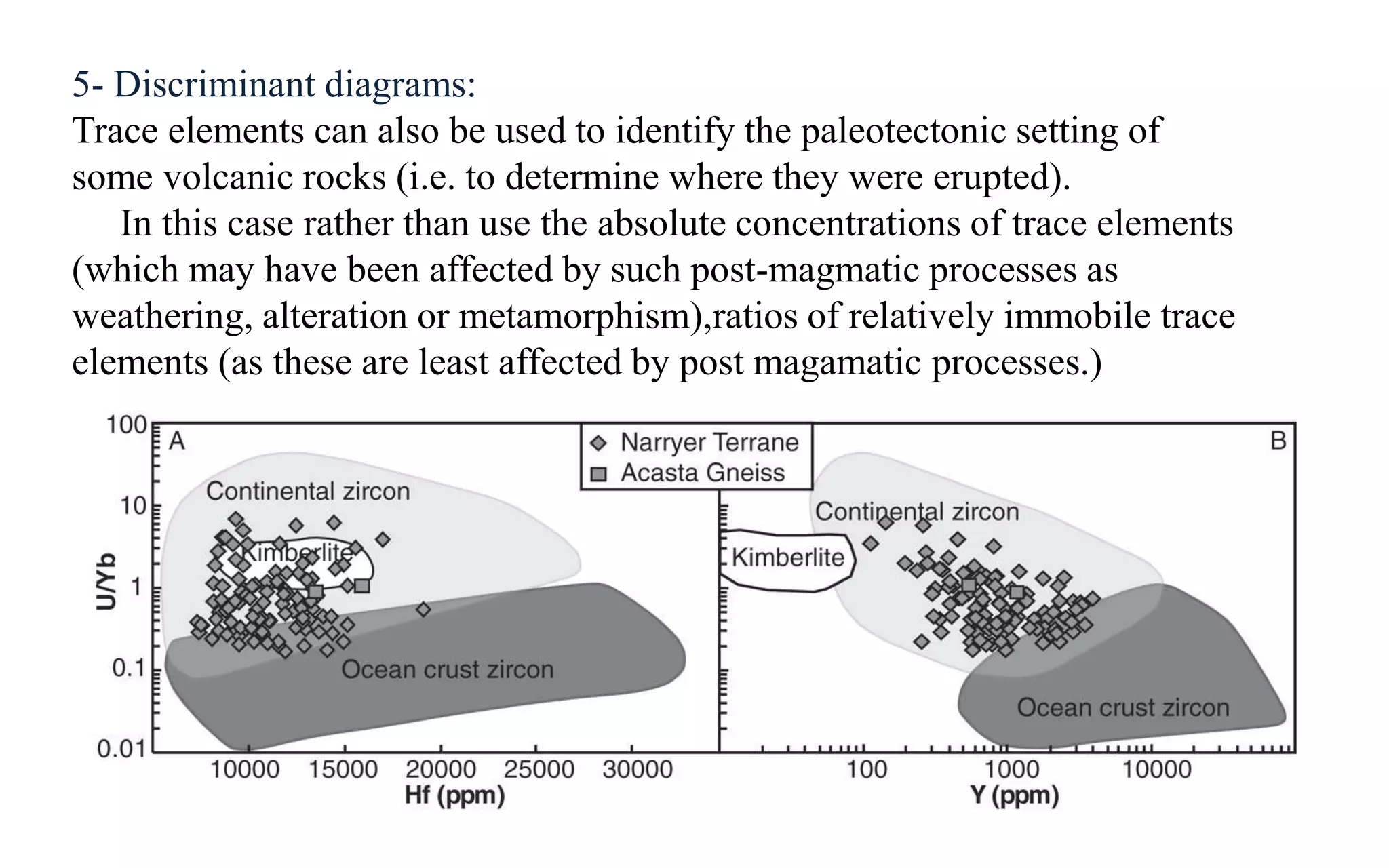
Trace elements occur in very low concentrations in rocks and provide important information about magmatic processes. They can be classified as compatible or incompatible based on whether they fit easily into mantle mineral crystal structures. Geochemical analysis of trace elements using techniques like XRF and ICP-MS allows determination of magma source and depth, identification of fractionating phases, and testing of models of magmatic differentiation. Trace elements are especially useful for rare earth elements, which indicate the type of basalt and can identify fractionating phases from REE patterns.