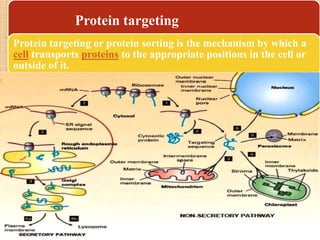
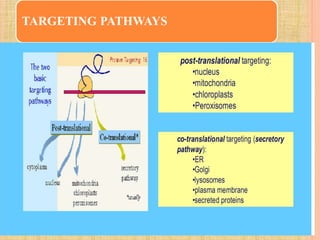
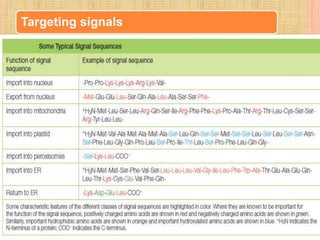
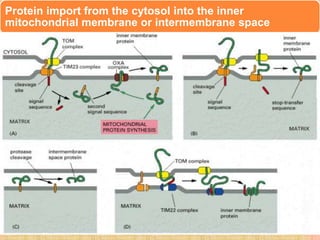
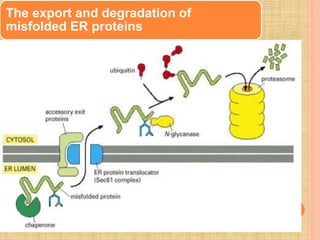
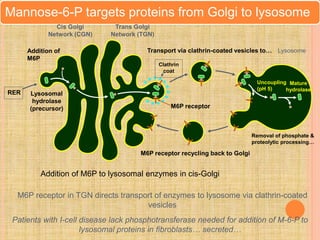
Protein sorting and targeting involves transporting proteins to the appropriate locations within or outside the cell. There are several pathways for protein targeting, including vesicular transport between organelles like the ER and Golgi, as well as transport of proteins into organelles like mitochondria and peroxisomes. Targeting signals like presequences and internal targeting peptides direct cellular transport machinery to correctly position proteins. Lipidation is another method to target proteins to specific membranes through modifications like glycosyl phosphatidylinositol anchors or myristoylation.

























![EUKARYOTIC PROTEIN TRANSPORT ACROSS MEMBRANES
Lipidation
Lipidation is a method to target proteins to membranes in organelles
(endoplasmic reticulum [ER], Golgi apparatus, mitochondria), vesicles
(endosomes, lysosomes) and the plasma membrane. The four types of
lipidation are:
C-terminal glycosyl phosphatidylinositol (GPI) anchor-
N-terminal myristoylation
S-myristoylation
S-prenylation
Each type of modification gives proteins distinct membrane affinities, although
all types of lipidation increase the hydrophobicity of a protein and thus its
affinity for membranes. The different types of lipidation are also not mutually
exclusive, in that two or more lipids can be attached to a given protein](https://image.slidesharecdn.com/proteintargetting-170703135712/85/Protein-targetting-32-320.jpg)