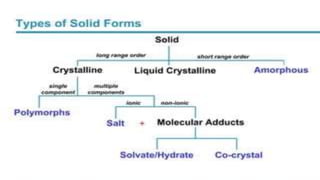
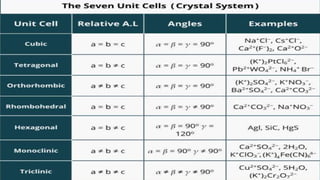
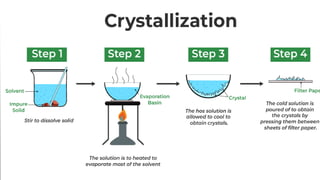
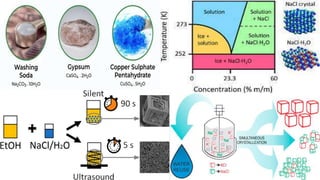
The document discusses crystallization as a solid-liquid separation technique essential for purifying solid compounds in various industries, particularly pharmaceuticals. It explains the processes and importance of crystallization from both aqueous and non-aqueous solutions, highlighting factors affecting crystal formation, such as solvent choice and environmental conditions. Additionally, it details the role of water of crystallization and factors impacting crystal morphology and growth rates.