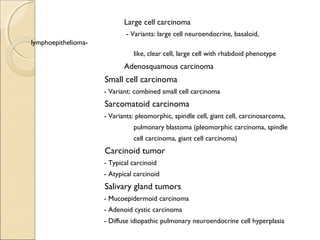
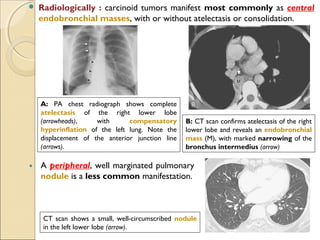
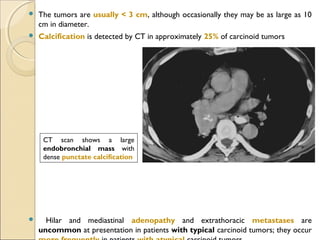
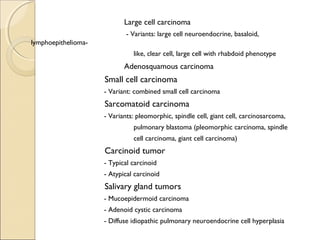
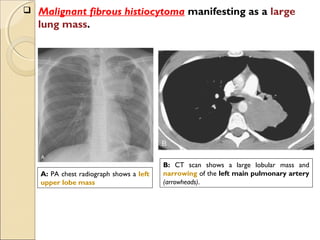
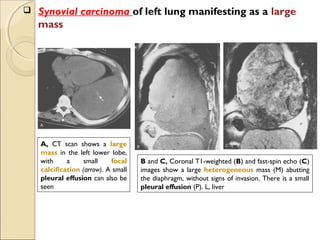
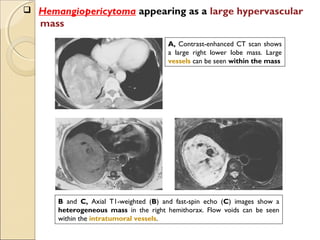
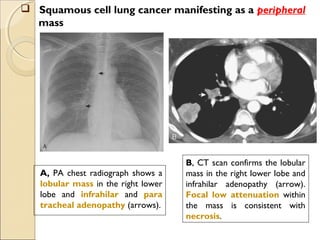
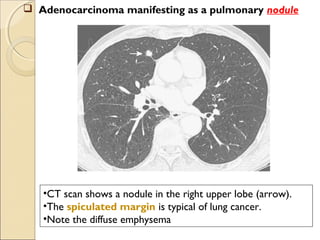
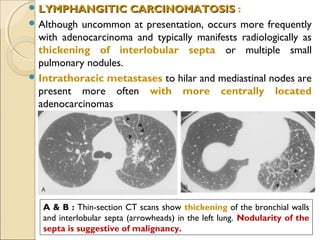
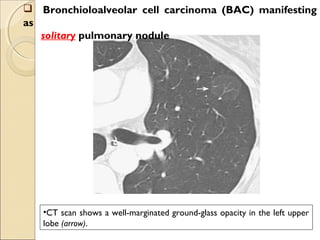
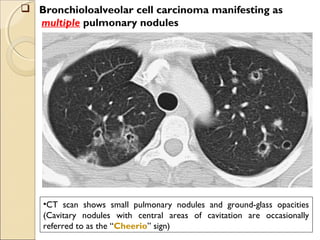
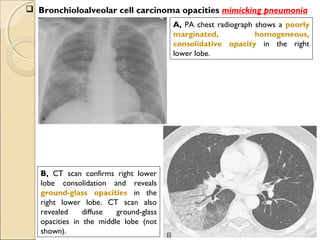
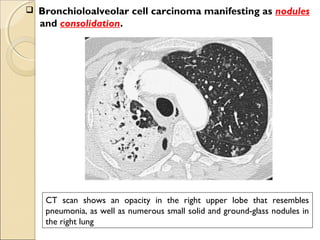
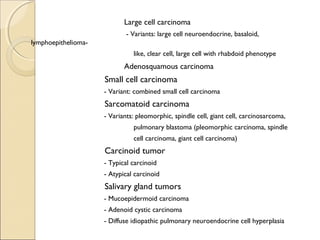
This document provides information on primary pulmonary neoplasms (lung cancer). It discusses the epidemiology and causes of lung cancer, including the major risk factor of cigarette smoking. It then covers the histologic classification of lung cancers, distinguishing between non-small cell lung carcinoma (NSCLC) and small cell lung carcinoma. Within NSCLC, it describes the characteristics and radiologic manifestations of the main subtypes: squamous cell carcinoma, adenocarcinoma, and other rare types. Key precursor lesions like atypical adenomatous hyperplasia and diffuse idiopathic pulmonary neuroendocrine cell hyperplasia are also summarized.




















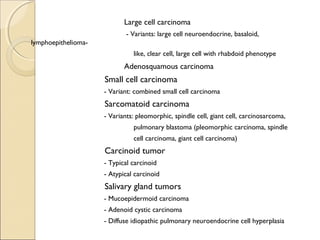
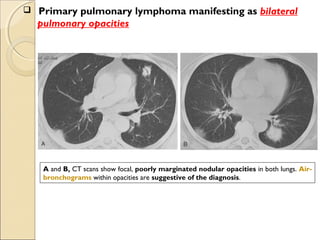
![ Non–small cell lung cancer in a 61-year-old woman presenting
with
hoarseness caused by involvement of the recurrent laryngeal nerve
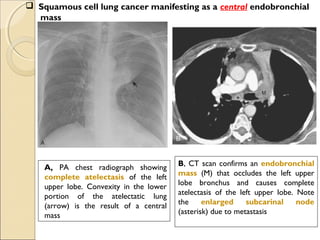
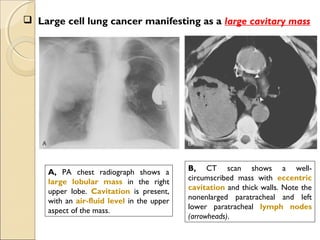
A, PA chest radiograph shows a left
perihilar mass (arrow).
[Note a small effusion and pneumothorax
following transthoracic needle aspiration
biopsy (arrowheads)]
B, CT scan reveals mediastinal
invasion, with extension of the
mass into the aortopulmonary
window. Arrows show the mass in
the recurrent laryngeal nerve.
(Note the small pleural effusion)](https://image.slidesharecdn.com/primarypulmonaryneoplasm-171226164010/85/Primary-pulmonary-neoplasm-37-320.jpg)


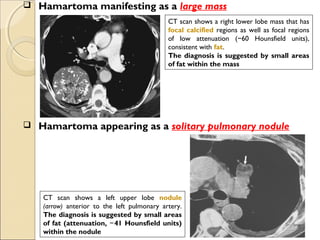
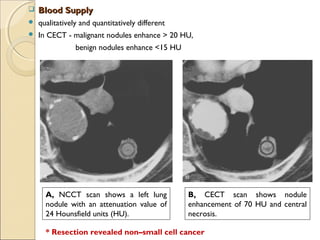
![ Internal Morphology.Internal Morphology.
Fat ( 40 to 120 [HU]) and− − calcification, only features that are reliable in
distinguishing lung cancer from a benign nodule.
Calcification in malignant lesions is typically amorphous in appearance (found in
up to 14% of lung cancers ).
Calcification in benign lesions is diffuse, solid, central punctate, laminated,
or popcorn-like.
Likelihood of malignancy varies according to the degree of soft tissue attenuation.
- partially solid nodular opacities (63%),
- ground-glass opacities (18%)
- solid nodules (7%)
Cavitation occurs in benign nodules and lung cancer.
Malignant nodules : typically thick, irregular walls,
Benign nodules : smooth, thin walls.
GrowthGrowth.
Lung cancers typically double in volume between 30 and 400 days (avg. 240 days)
The measurement of serial volumes, rather than diameters, and the computer-
calculated doubling of volume of small nodules have been suggested as accurate and
potentially useful methods to assess growth](https://image.slidesharecdn.com/primarypulmonaryneoplasm-171226164010/85/Primary-pulmonary-neoplasm-43-320.jpg)

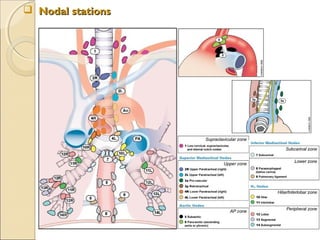
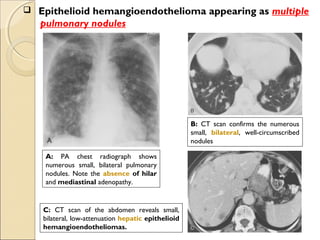
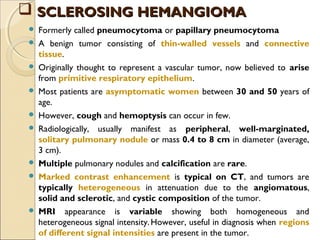
![ International Staging System for Lung CancerInternational Staging System for Lung Cancer
Primary Tumor (T)Primary Tumor (T)
TxTx Primary tumor cannot be assessed, or tumor proved by the presence of malignant cells in sputum or bronchial washings
but not visualized by imaging or bronchoscopy
T0T0 No evidence of primary tumor
TisTis Carcinoma in situ
T1T1 Tumor ≤3 cm in greatest dimension, surrounded by lung or visceral pleura, without bronchoscopic evidence of invasion
more proximal than the lobar bronchus[*]
(i.e., not in the main bronchus)
T2T2 Tumor with any of the following features of size or extent: >3 cm in greatest dimension; Involves main bronchus, ≥2 cm
distal to the carina; Invades the visceral pleura; Associated with atelectasis or obstructive pneumonitis that extends to
the hilar region but does not involve the entire lung
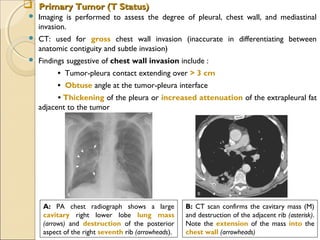
T3T3 Tumor of any size that directly invades any of the following: chest wall (including superior sulcus tumors), diaphragm,
mediastinal pleura, or parietal pericardium; or tumor in the main bronchus <2 cm distal to the carina, but without
involvement of the carina; or associated atelectasis or obstructive pneumonitis of the entire lung
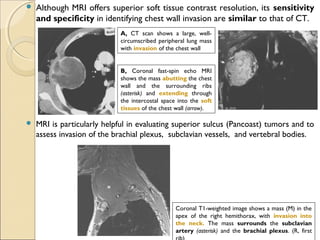
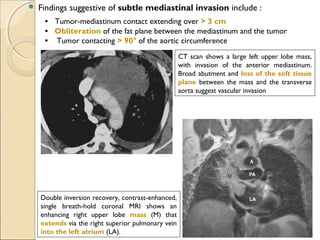
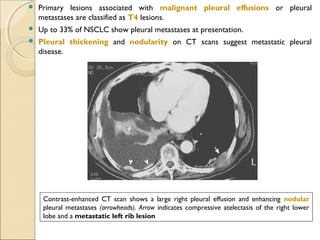
T4T4 Tumor of any size that invades any of the following: mediastinum, heart, great vessels, trachea, esophagus, vertebral
body, or carina; or tumor with a malignant pleural or pericardial effusion[†]
or with satellite tumor nodule(s) within the
ipsilateral primary-tumor lobe of the lung
Regional Lymph Nodes (N)Regional Lymph Nodes (N)
NxNx Regional lymph nodes cannot be assessed
N0N0 No evidence of regional lymph node metastasis
N1N1 Metastasis to : ipsilateral peribronchial and/or ipsilateral hilar lymph nodes
: intrapulmonary nodes involved by direct extension of the primary tumor
N2N2 Metastasis to : ipsilateral mediastinal and/or subcarinal lymph node(s)
N3N3 Metastasis to : contralateral mediastinal or hilar lymph node(s)
: ipsilateral or contralateral scalene, or supraclavicular lymph node(s)
Distant Metastasis (M)Distant Metastasis (M)
MxMx Presence of distant metastasis cannot be assessed
M0M0 No distant metastasis
M1M1 Distant metastasis present[‡]
* Superficial tumor of any size limited to the bronchial wall, is also classified as T1.
† In a few patients, cytopathologic examinations of pleural/pericardial fluid show no tumor, is nonbloody and non-exudative. Then it should be
excluded as a staging element, and the patient's disease should be staged T1, T2, or T3.
‡ Separate metastatic tumor nodule(s) in the ipsilateral nonprimary-tumor lobe(s) of the lung are also classified M1.](https://image.slidesharecdn.com/primarypulmonaryneoplasm-171226164010/85/Primary-pulmonary-neoplasm-48-320.jpg)