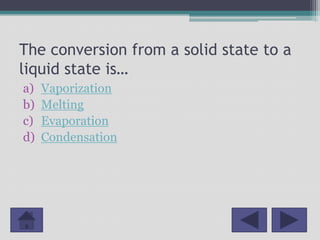
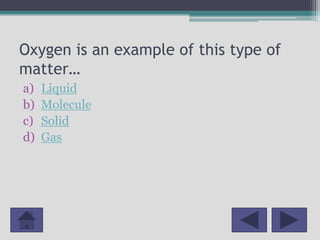
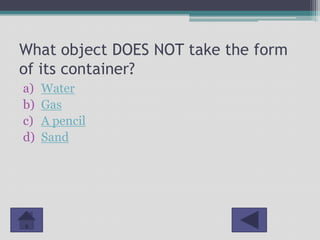
This document outlines a lesson plan for 2nd grade science focused on the states of matter: solids, liquids, and gases. It includes curriculum objectives, interactive activities for students to classify and observe matter, and key concepts related to the properties of different states. Additionally, it provides resources and references for further education on the topic.