Embed presentation
Downloaded 12 times



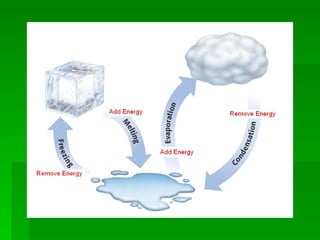






The document discusses different changes of state that substances can undergo. It explains that changing from one state to another, such as solid to liquid, requires adding or removing energy. Melting occurs when a solid turns to a liquid, and freezing is the reverse change. Evaporation changes a liquid to a gas below the boiling point, while boiling occurs when a liquid turns to a gas throughout. Lower pressure lowers the boiling point of a substance, while higher pressure raises the boiling point. The document also mentions sublimation, which is when a solid turns directly to a gas.









