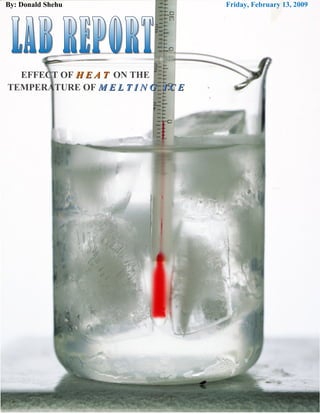
The document describes an experiment to observe the temperature changes that occur when ice melts. Key findings include:
- The temperature slowly increases as the ice melts, absorbing the heat from the Bunsen burner.
- The heat causes the ice particles to vibrate faster until melting occurs at 0°C, the melting point of ice.
- At the melting point, the temperature remains constant until all the ice is transformed into liquid water.