Report
Share
Recommended
Recommended
Matter in our surroundings

CHANGE
OF
STATE
STATES OF MATTER
There are three states, or phases, of matter.
They are:
Solid
Liquid
Gas
When a substance changes from one state, or phase, of matter to another we say that it has undergone a change of state.
MELTING
The change of state from solid to liquid state is called melting.
The temperature at which the solid states of the substance changes into the liquid states of the substance is called its melting point.
The temperature remains constant until the entire amount solid is converted into liquid state
VAPORISATION
The change of state from liquid to vapour is called vaporisation.
The liquid and vapour states of the substance coexist in thermal equilibrium during the change of state from liquid to vapour.
The temperature at which the liquid states of the substance change to its vapour state is called its boiling point.
CONDENSATION
The process of changing gas (or vapour) to a liquid by cooling is called condensation
Condensation is the reverse of boiling (or vaporisation).
FREEZING
The process f changing a liquid into a solid by cooling is called freezing
Freezing is reverse of melting
Freezing means solidification.
SUBLIMATION AND DESPOSITION
The process of changing of solid directly into vapour (or gas) is called sublimation.
The process of changing of vapour (or gas) directly into solid is called sublimation and desposition.
EFFECT OF PRESSURE ON MELTING POINT
Under normal atmospheric pressure, ice melts at 0 °C. However, this can change if the air pressure changes.
When the two ice cubes are pressed together, the melting point is lowered.
The ice melts since its temperature (0 °C) is above the new melting point.
When the ice cubes are released, the ice freezes again at 0 °C joining the two ice cubes together.
Presentation Matter In Our Surroundings

PPT on matter in our surroundings chapter1 for the class 9th ncert books
Matter in our surroundings_Class 9 CBSE

Complete, in detail, information about chapter 1 of class 9 CBSE board.
More Related Content
What's hot
Matter in our surroundings

CHANGE
OF
STATE
STATES OF MATTER
There are three states, or phases, of matter.
They are:
Solid
Liquid
Gas
When a substance changes from one state, or phase, of matter to another we say that it has undergone a change of state.
MELTING
The change of state from solid to liquid state is called melting.
The temperature at which the solid states of the substance changes into the liquid states of the substance is called its melting point.
The temperature remains constant until the entire amount solid is converted into liquid state
VAPORISATION
The change of state from liquid to vapour is called vaporisation.
The liquid and vapour states of the substance coexist in thermal equilibrium during the change of state from liquid to vapour.
The temperature at which the liquid states of the substance change to its vapour state is called its boiling point.
CONDENSATION
The process of changing gas (or vapour) to a liquid by cooling is called condensation
Condensation is the reverse of boiling (or vaporisation).
FREEZING
The process f changing a liquid into a solid by cooling is called freezing
Freezing is reverse of melting
Freezing means solidification.
SUBLIMATION AND DESPOSITION
The process of changing of solid directly into vapour (or gas) is called sublimation.
The process of changing of vapour (or gas) directly into solid is called sublimation and desposition.
EFFECT OF PRESSURE ON MELTING POINT
Under normal atmospheric pressure, ice melts at 0 °C. However, this can change if the air pressure changes.
When the two ice cubes are pressed together, the melting point is lowered.
The ice melts since its temperature (0 °C) is above the new melting point.
When the ice cubes are released, the ice freezes again at 0 °C joining the two ice cubes together.
Presentation Matter In Our Surroundings

PPT on matter in our surroundings chapter1 for the class 9th ncert books
Matter in our surroundings_Class 9 CBSE

Complete, in detail, information about chapter 1 of class 9 CBSE board.
What's hot (19)
Similar to Matter (lesson 3) changes of state in matter
Changes around us

This PPT prepared by student-Teacher from Thiagarajar College of Preceptors for Demo Class
Changes in matter.pptx 20013 2014 ( Physical and chemical change)

Changes in matter ( physical and chemical change)
phasechanges.pptx

Notes 2: Phase Change
• Phase change is a change from one state of matter (solid, liquid, gas) to another.
• Phase changes are physical changes because it only affects physical appearance, not chemical make-up and its reversible
• Water exists on our planet in three states: Ice, water, and water vapor
• Water is different from other substances because it is less dense as a solid than liquid
• When temperature changes, molecules speed up/slow down resulting in a change in the average kinetic energy of molecules.
• During a phase change, heat energy is either absorbed or released. Heat energy is released as molecules move closer together. Heat energy is absorbed as molecules move farther apart.
• During phase changes, temperature does not change. Molecules move closer together or farther apart, resulting in a change in the potential energy of the molecules.
• Types of phase changes: Melting, freezing, vaporization, condensation, sublimation, deposition
• Melting - Phase change from a solid to a liquid
• Freezing- Phase Change from a liquid to a solid
• Vaporization - Phase change from a solid or liquid to gas. It occurs at the boiling point of matter or sublimation process. (Water - 100° C, Table salt - 1413° C, Diamond - 4200° C)
• Evaporation-type of vaporization in which a liquid turns to a gas on the surface of a liquid (occurs at all temperatures).
• Evaporation- a Cooling Process. As water in the perspiration evaporates from your skin it absorbs and carries away heat energy from your body.
• Condensation - Phase change from a gas to a liquid.
• Water vapor in surrounding air loses heat energy when it comes in contact with the cold glass. Water vapor condenses and becomes liquid drops of water.
• Sublimation - Phase change from a solid to a gas.
• Deposition - Phase change from a gas to a solid.
• Boiling Point - The temperature at which a liquid changes into a gas. 212 F or 100 C
• Melting Point - The temperature at which a solid changes into a liquid. 32 F or 0 C
• Freezing point - The temperature at which a liquid changes into a solid. 32 F or 0 C
• Freezing point and melting point of a substance are the same temperature.
MATTER IN OUR SURROUNDING

Matter- Matter is anything which occupies space and has mass is called matter. Air and water, sugar and sand, hydrogen and oxygen etc. Matter is made up of very small tiny particles. Particles of matter have space between them they attract each other.
CHANGES AROUND US.pptx for class 7 , 8,9 and 10 for cbse students

Changes around us geography lesson for classes 10,9,8,7 and 6 for cbse and matriculation students
Changes in Matter Education Presentation in Orange Light Yellow Hand Drawn St...

Change of Object in our life
Changing States of Matter for Middle School

This Changing States of Matter slideshow for middle school students is ideally used to teach students about the various processes that change one state of matter to another. The processes featured are freezing, melting, evaporation, condensation, sublimation, and deposition. Thermal energy and movement of molecules are discussed.
To download slideshow or the powerpoint version of this slideshow, visit The Homeschool Daily. Be sure to check out our full line of homeschool curriculum. Thanks for looking!
Similar to Matter (lesson 3) changes of state in matter (20)
Changes in matter.pptx 20013 2014 ( Physical and chemical change)

Changes in matter.pptx 20013 2014 ( Physical and chemical change)
CHANGES AROUND US.pptx for class 7 , 8,9 and 10 for cbse students

CHANGES AROUND US.pptx for class 7 , 8,9 and 10 for cbse students
Changes in Matter Education Presentation in Orange Light Yellow Hand Drawn St...

Changes in Matter Education Presentation in Orange Light Yellow Hand Drawn St...
Matter (lesson 3) changes of state in matter
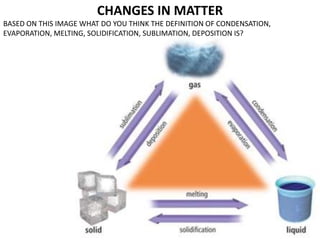
- 1. CHANGES IN MATTER BASED ON THIS IMAGE WHAT DO YOU THINK THE DEFINITION OF CONDENSATION, EVAPORATION, MELTING, SOLIDIFICATION, SUBLIMATION, DEPOSITION IS?
- 2. If we melt an ice cube can it be frozen again? Yes, this is called reversing changes in physical matter We can also reverse changes in physical matter by: • A bottle of frozen water thawed to a liquid state again • Water vapour that has condensed on a cold window evaporates into a vaporous state again • water from a puddle that has evaporated will fall to the ground as rain
- 3. Chemical changes are irreversible • when the chrome on a bicycle rusts, it can never go back to being chrome • when an egg is boiled it can never go back to being a raw egg
- 4. What happens when water freezes? • when water freezes it releases heat What happens when an when an ice cube melts? • it absorbs heat This means that changes of state involve the release of heat or the absorption of heat