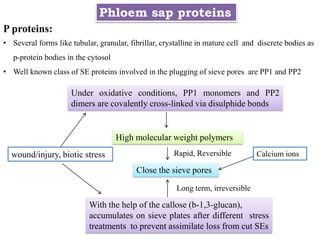
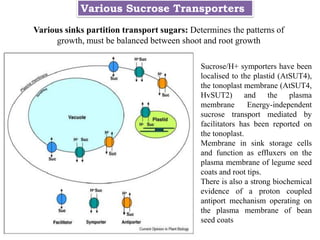
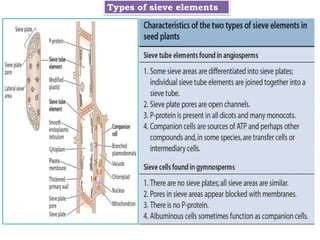
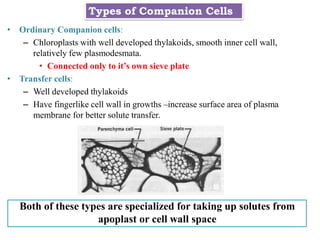
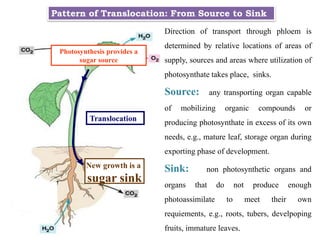
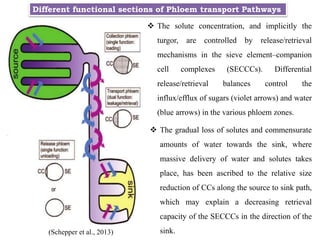
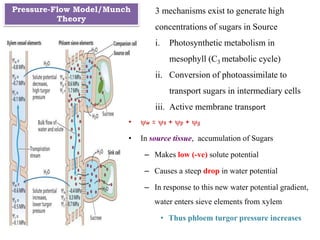
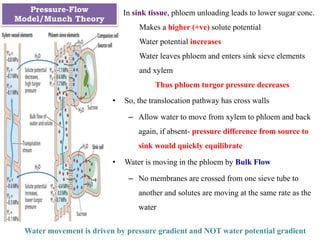
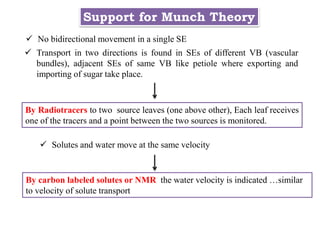
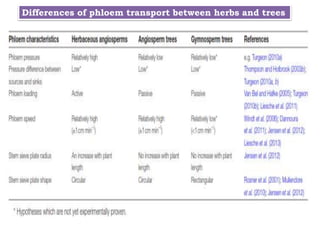
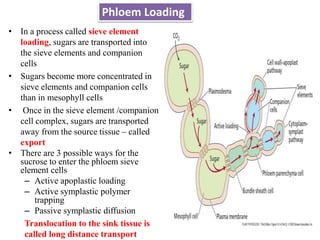
The document provides an extensive overview of phloem translocation, focusing on the anatomy of phloem, specifically sieve elements and companion cells, along with the mechanisms of translocation from source to sink. It discusses loading strategies, the composition of phloem sap, and the pressure-flow model while outlining the differences between various types of companion cells and translocational pathways. The document also addresses factors affecting translocation patterns and experimental methods used to understand phloem transport dynamics.
















![ Phloem sap is rich in organic molecules and ions…needs a
elaborated set of transporters unless the composition of sap
changes at each step.
Anatomical studies of Phaseolus vulgaris do not support the
hypothesis
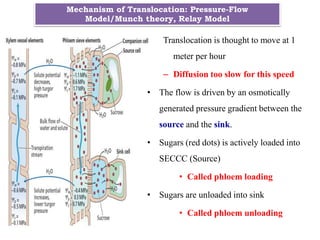
Resistance in transport phloem is proportional to the plant length,
the resistances in the Collection and Release phloem are inversely
proportional to the source (leaf length) and sink (root length).
Altered relationship of resistance for Relay Model.
Difficulties of Relay Model
R3α Ll x S l (For herbs and trees) [R= sieve tube radius, Ll= leaf length, Sl= Stem length]](https://image.slidesharecdn.com/phloemtransportm-200328164959/85/Phloem-transport-m-sc-c-u-25-320.jpg)

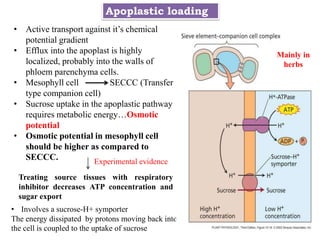




![• Apoplastic: three types
• 1 [B] One step, transport from the sieve element-companion cell complex to
successive sink cells, occurs in the apoplast.
• Once sugars are taken back into the symplast of adjoining cells transport is
symplastic
Apoplastic phloem loading](https://image.slidesharecdn.com/phloemtransportm-200328164959/85/Phloem-transport-m-sc-c-u-35-320.jpg)
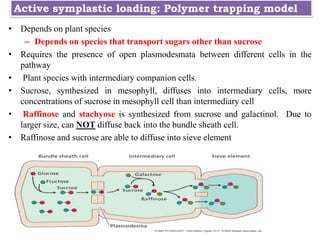
![(2) [A] involves an apoplastic step close to the sieve element
companion cell.
(2) [B] involves an apoplastic step which is farther removed from the
SECCs (In seeds sucrose transported maternal tissue to embryo)
Both involve movement through the plant cell wall](https://image.slidesharecdn.com/phloemtransportm-200328164959/85/Phloem-transport-m-sc-c-u-36-320.jpg)