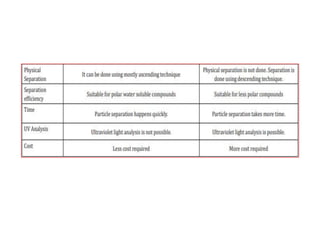
This document discusses paper chromatography and thin layer chromatography. Paper chromatography uses cellulose filter paper as the stationary phase and a liquid as the mobile phase to separate solid and liquid compounds based on their polarity. Thin layer chromatography uses a silica gel coated glass plate as the stationary phase and a liquid mobile phase to separate non-volatile mixtures based on the polarity of particles toward the phases. Both techniques work on the principle of separating compounds based on their distribution between a solid stationary phase and liquid mobile phase.