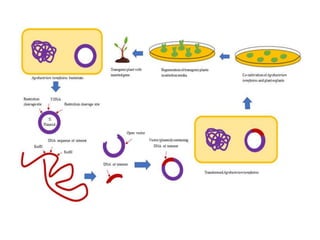
Agro-bacterium-mediated transformation is a common method for inserting genes into dicotyledonous plants using the soil bacterium Agro-bacterium. The method involves using Agro-bacterium containing a T-DNA vector to transfer genes of interest into plant cells, followed by regeneration of transformed plants on selection media. Protocols are provided for transforming tobacco and chrysanthemum explants using Agro-bacterium, including co-cultivation, selection, regeneration of shoots, rooting of shoots, and verification of transformation. Transformed tobacco and chrysanthemum plants were successfully recovered using this method.