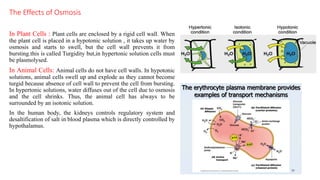
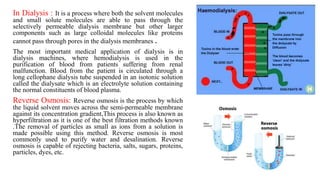
The document discusses the role of osmosis in animal cells, explaining its definition, types of solutions, and implications for cell stability. It emphasizes the importance of maintaining an isotonic environment for animal cells to prevent osmotic lysis and describes the consequences of different solutions on cell behavior. Additionally, it covers related topics such as dialysis, reverse osmosis, and the significance of osmosis in physiological processes and drug delivery systems.