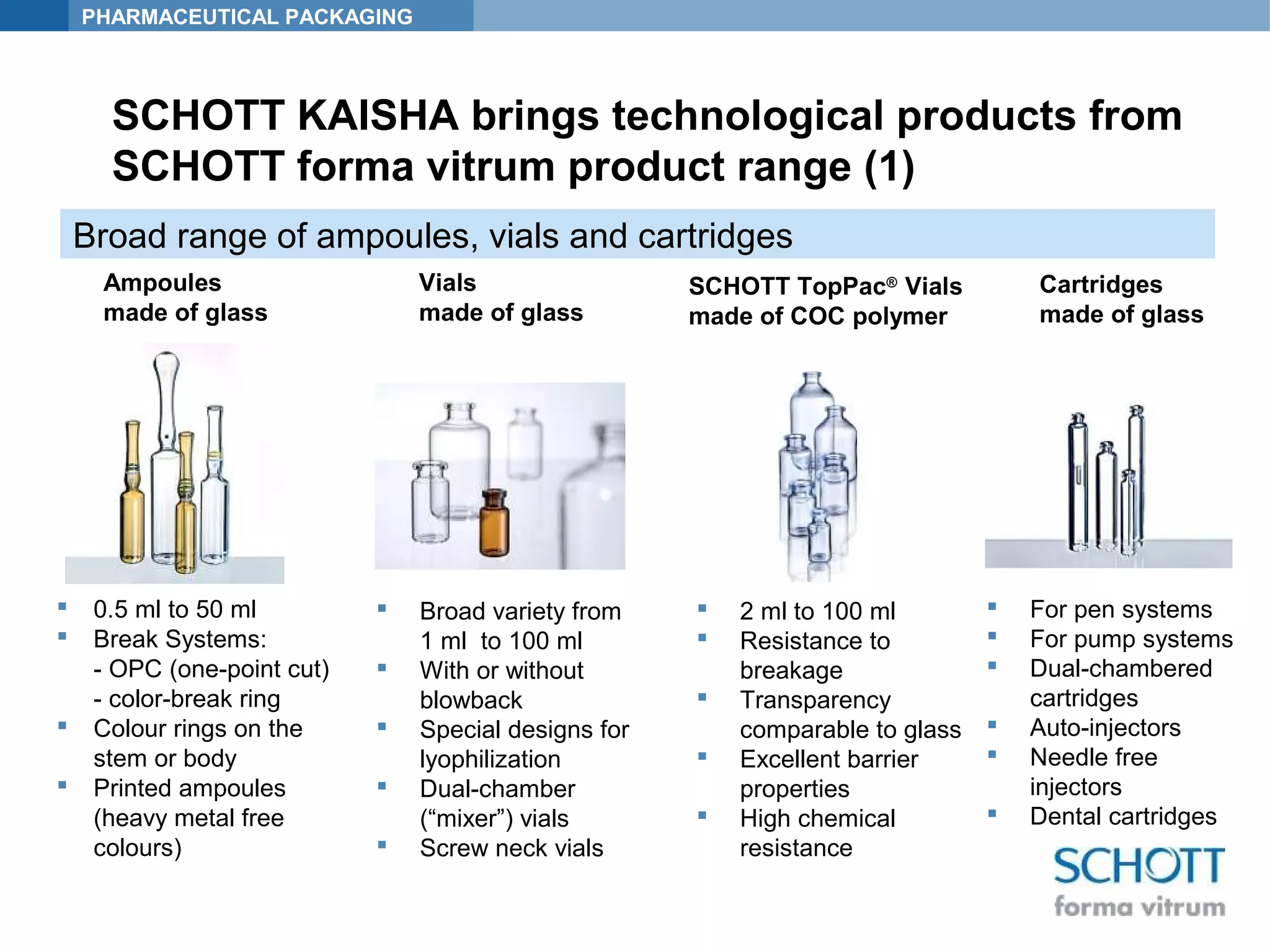
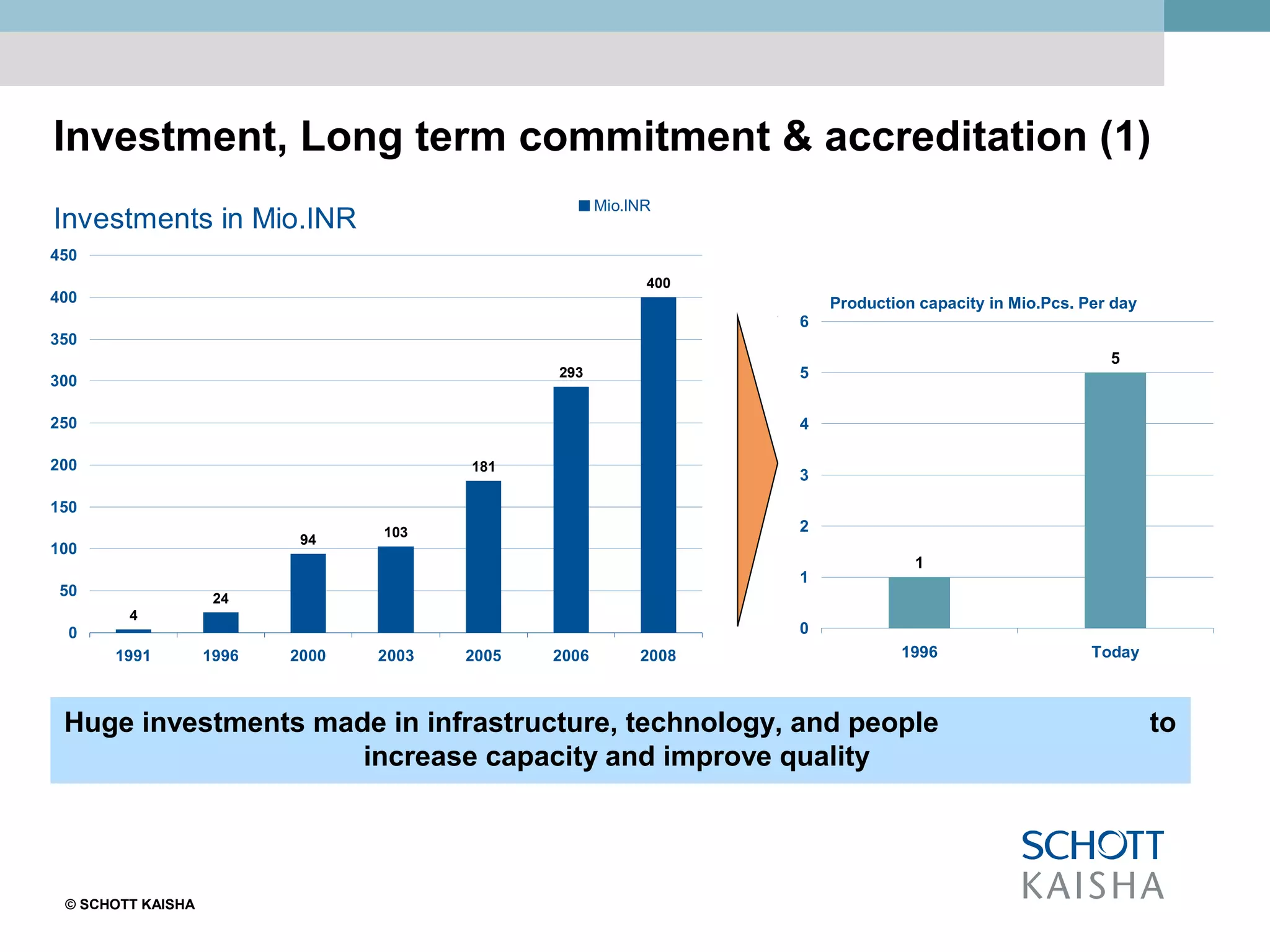
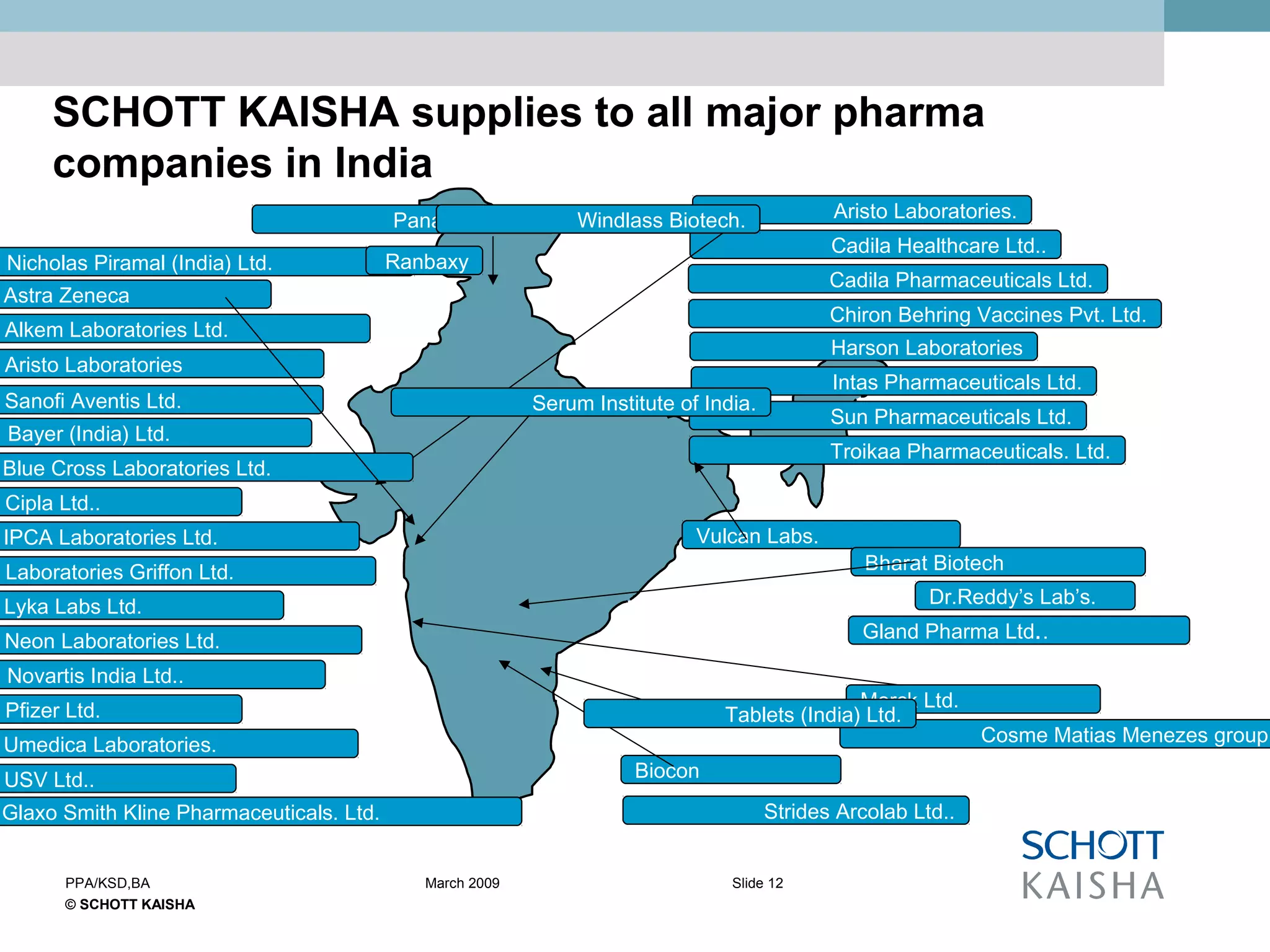
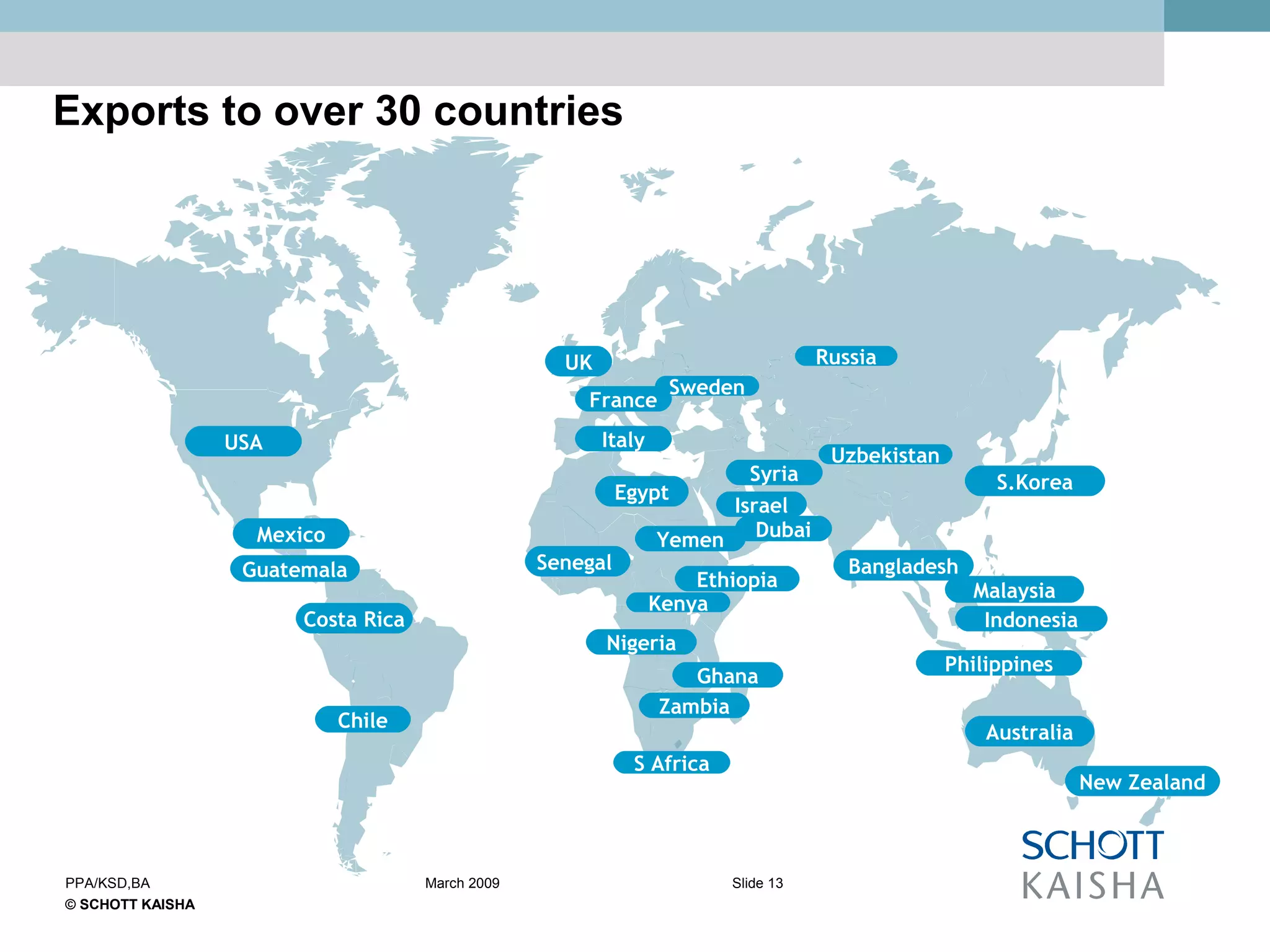
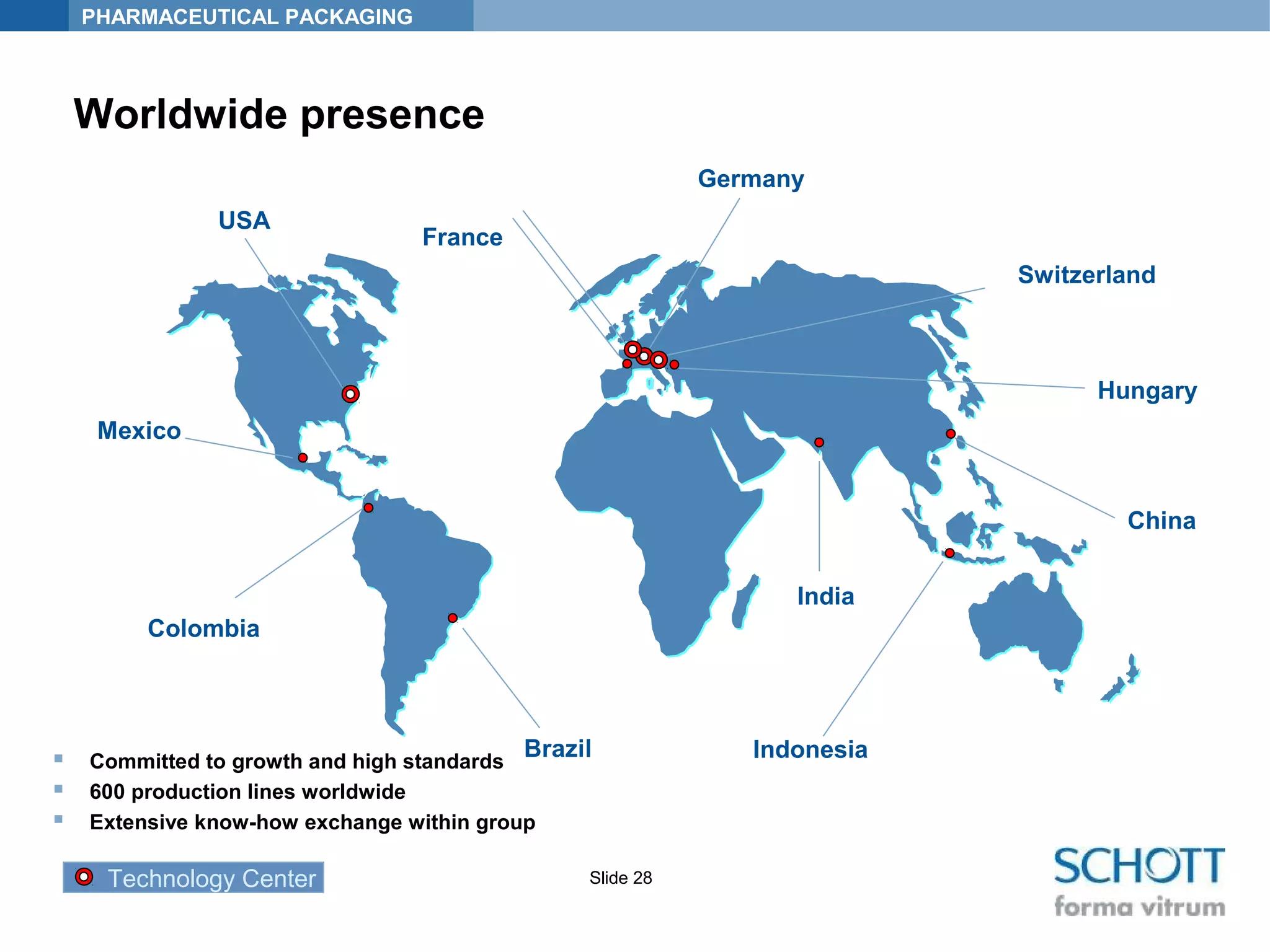
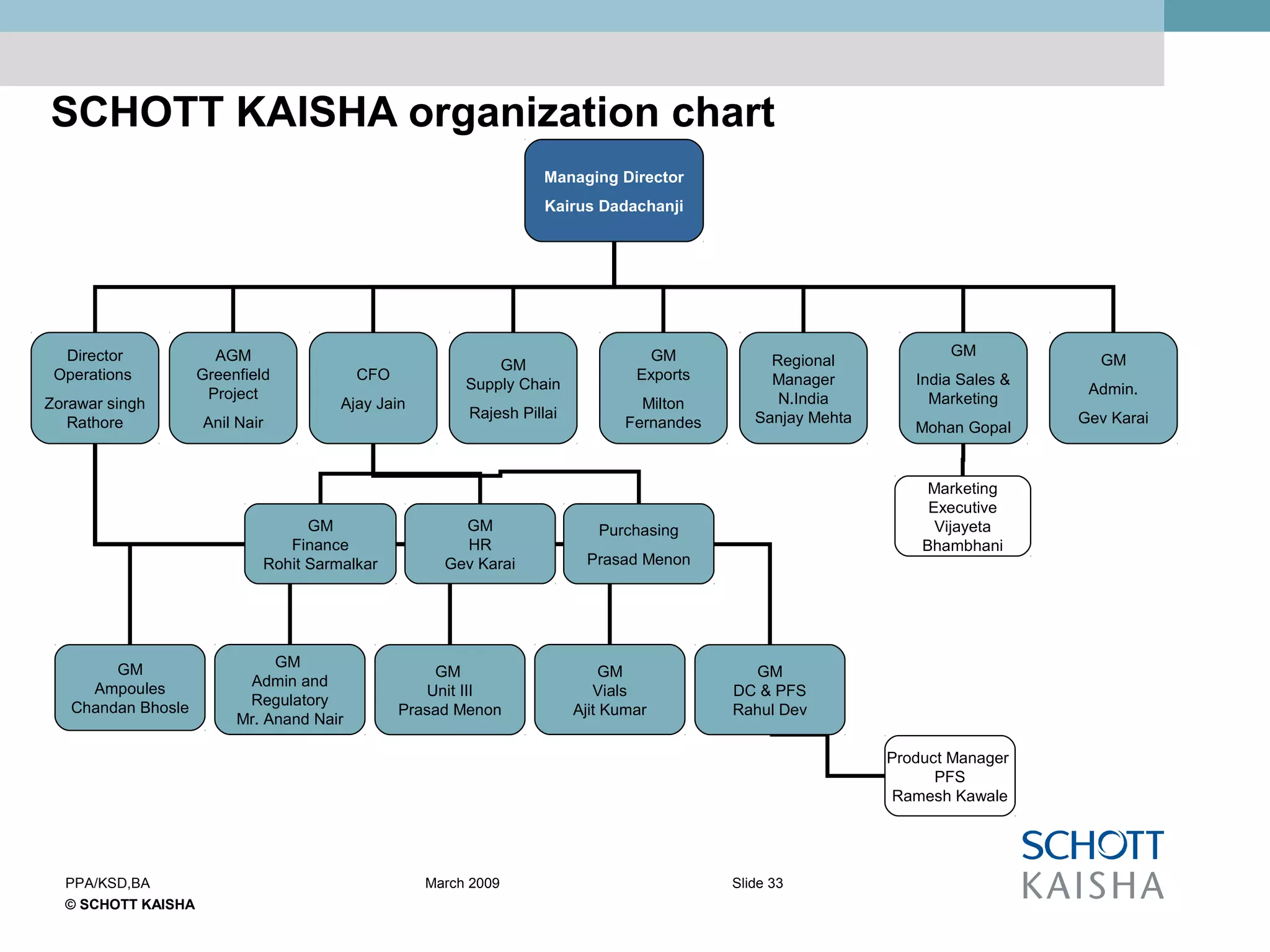
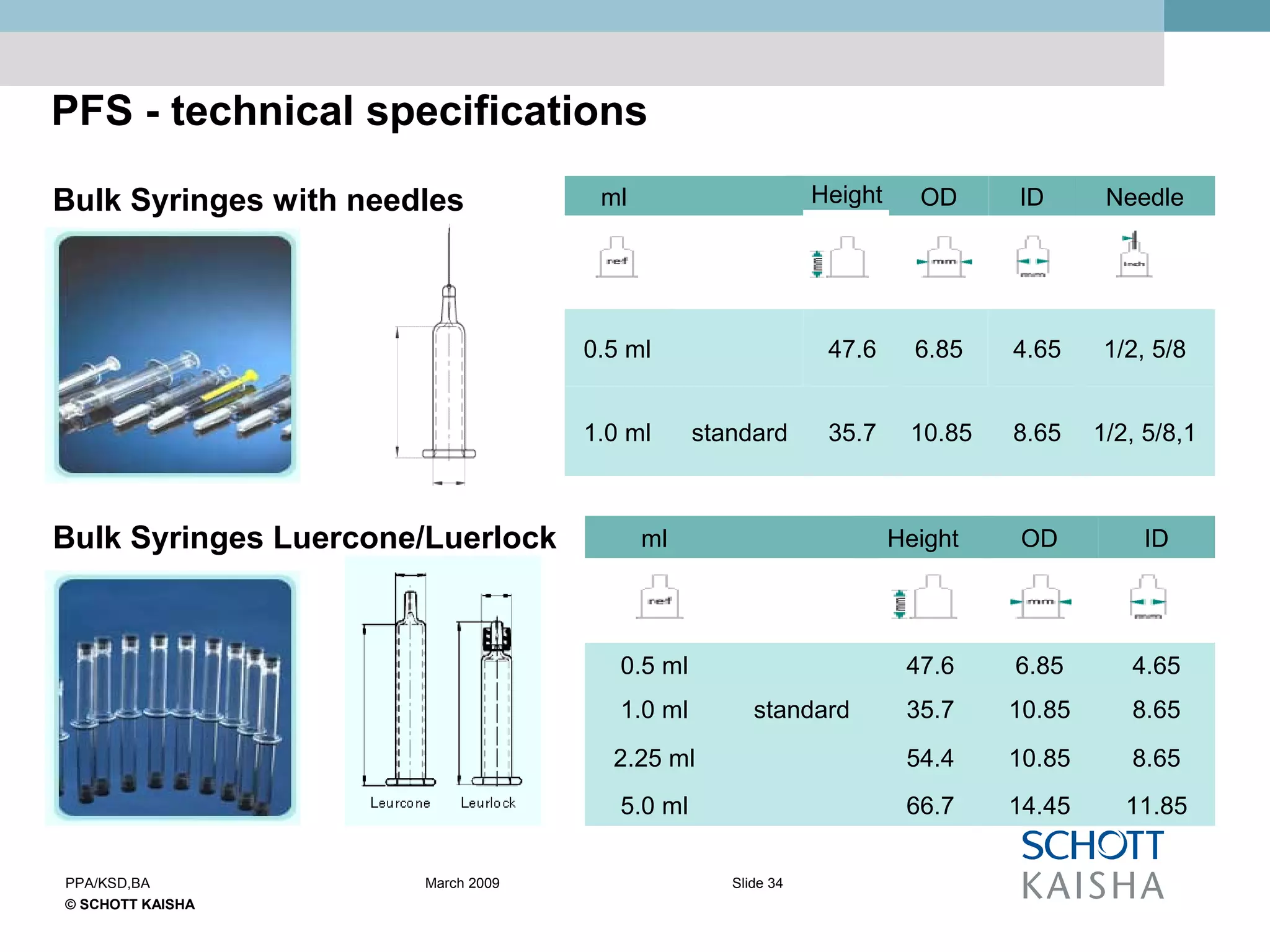
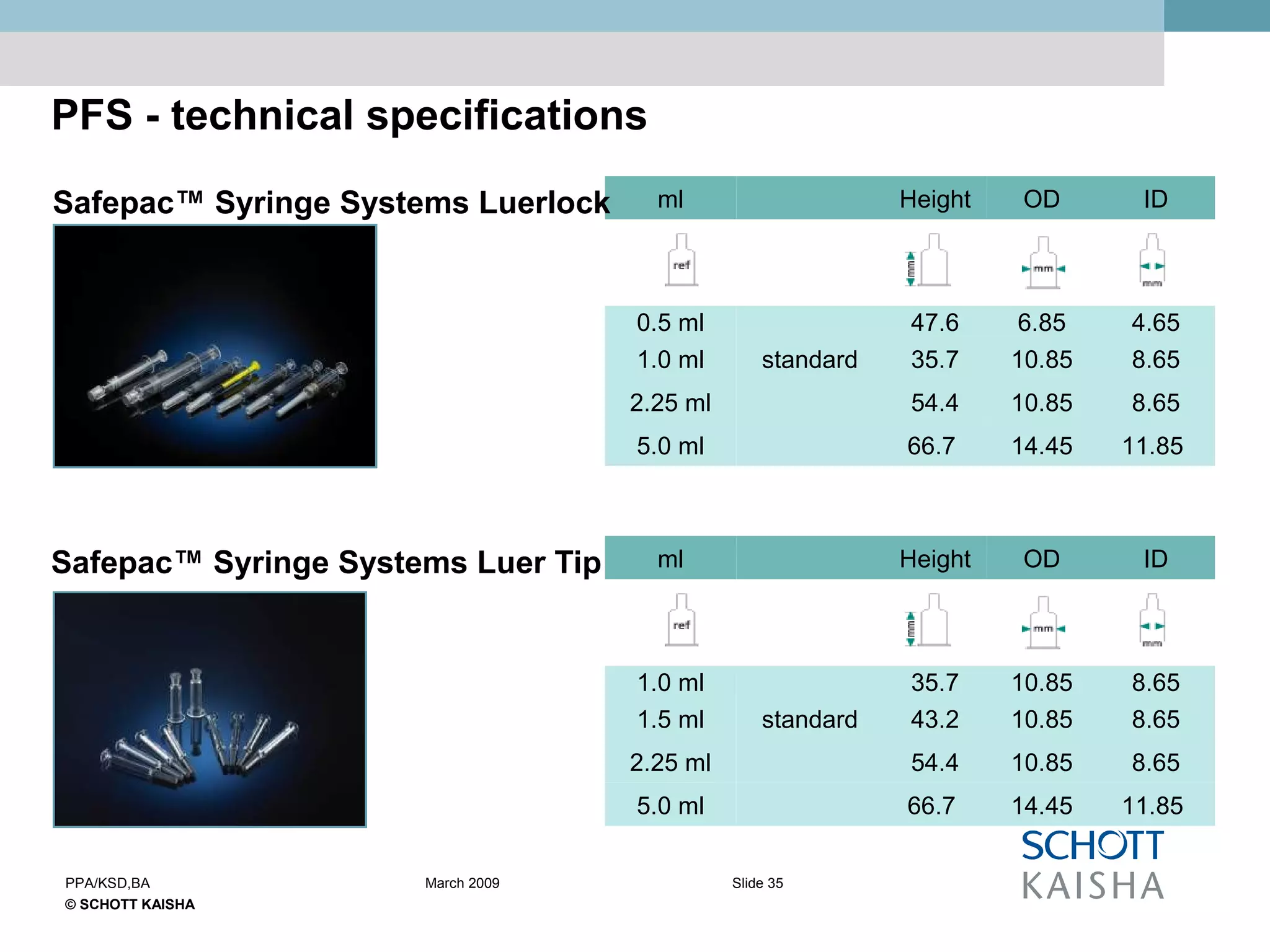
SCHOTT KAISHA is a joint venture between SCHOTT forma vitrum, a global leader in pharmaceutical glass packaging, and an Indian company. It has made large investments over time to expand production capacity of products like ampoules, vials, and syringes. SCHOTT KAISHA supplies major Indian pharmaceutical companies and exports to over 30 countries. It aims to provide a one-stop shop for customers' primary and secondary packaging needs through its partnership with SCHOTT and adherence to high quality and environmental standards.