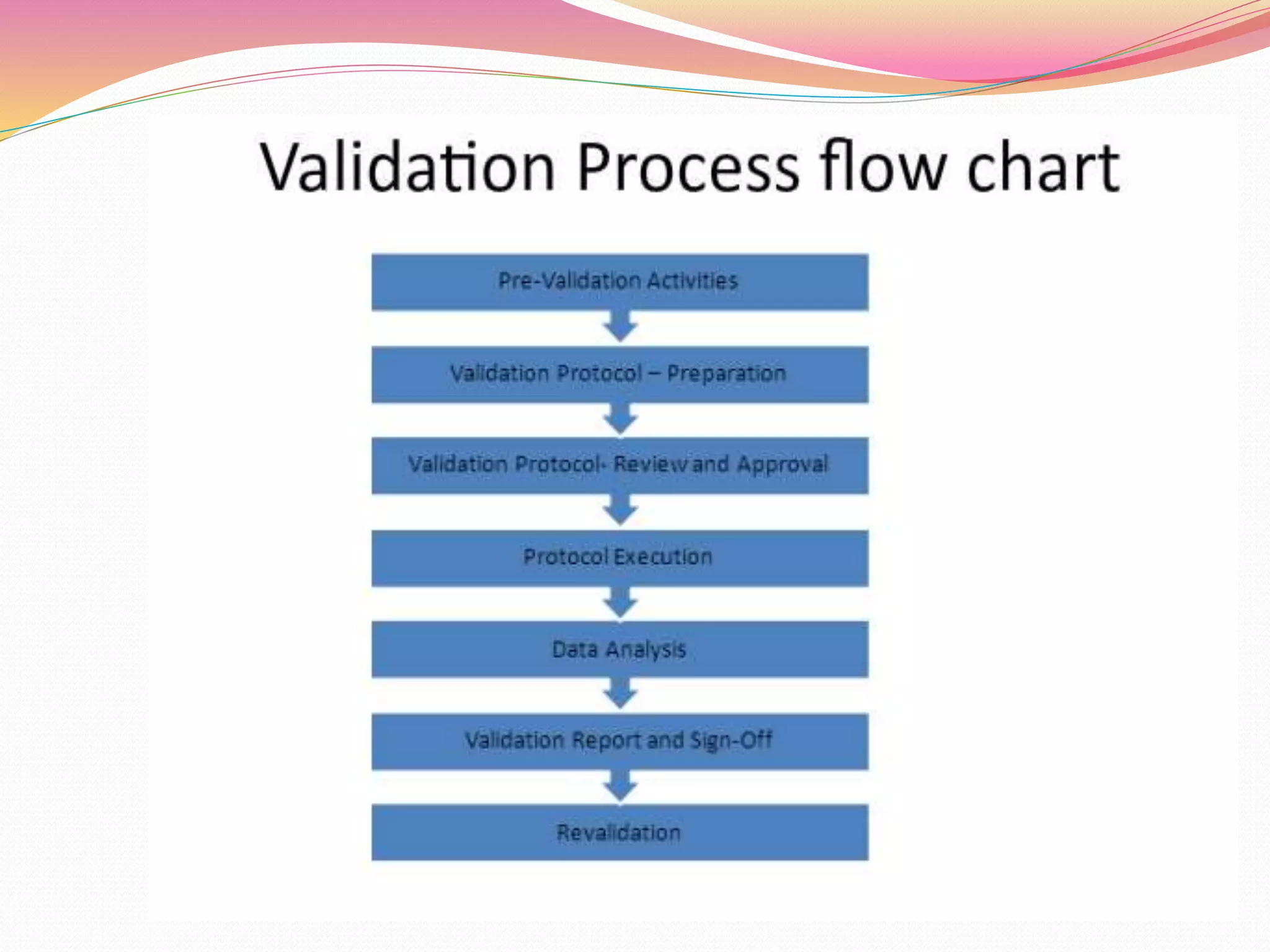
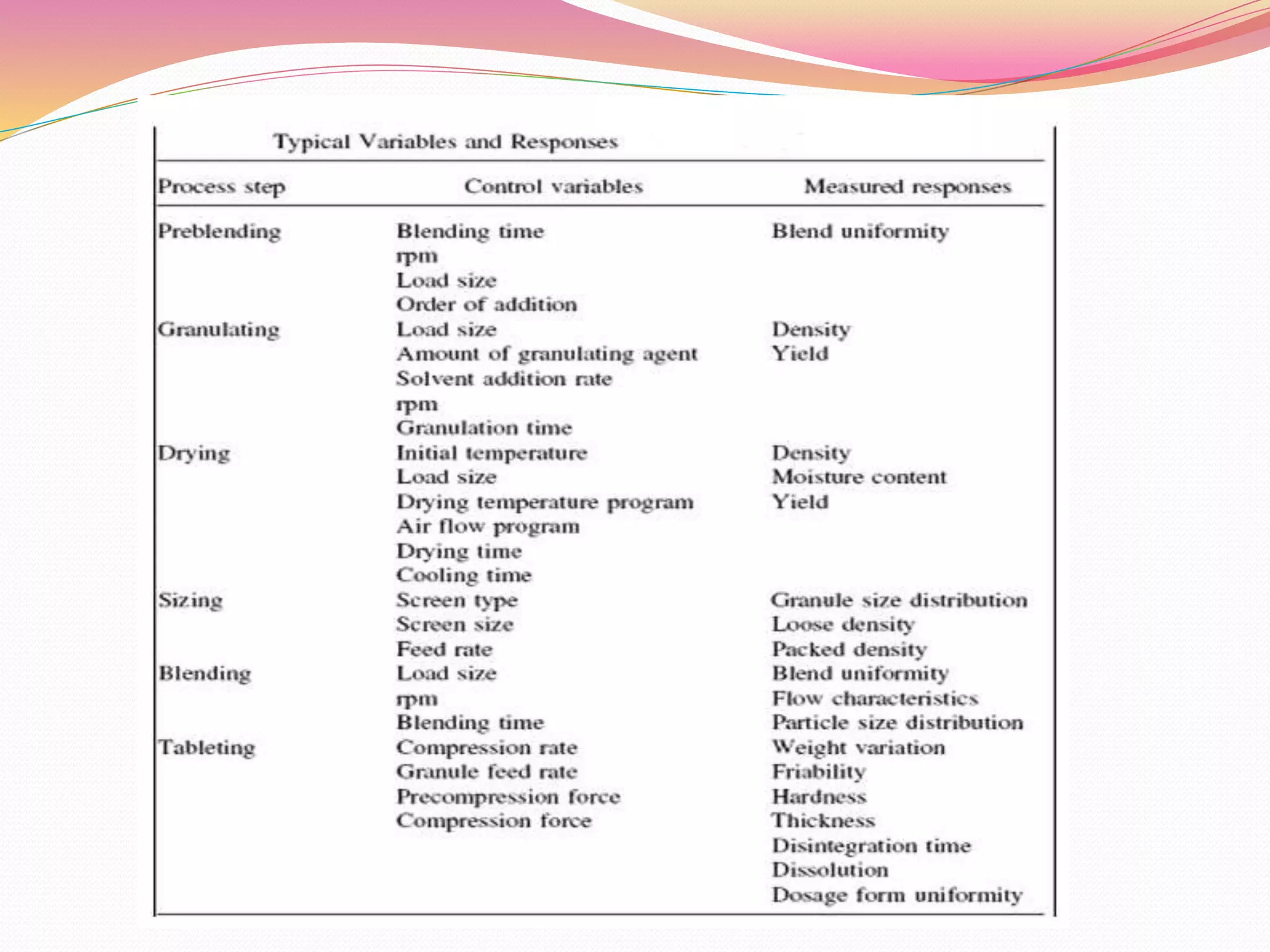
The document discusses validation methods for equipment and processing techniques in pharmaceutical manufacturing, focusing on various aspects such as mixing, granulation, drying, compression, filtration, and filling. It outlines the importance of process validation to ensure that production methods consistently meet predefined specifications and quality attributes. Additionally, it describes the qualification processes for equipment and the significance of a multidisciplinary validation team in maintaining quality and regulatory compliance.