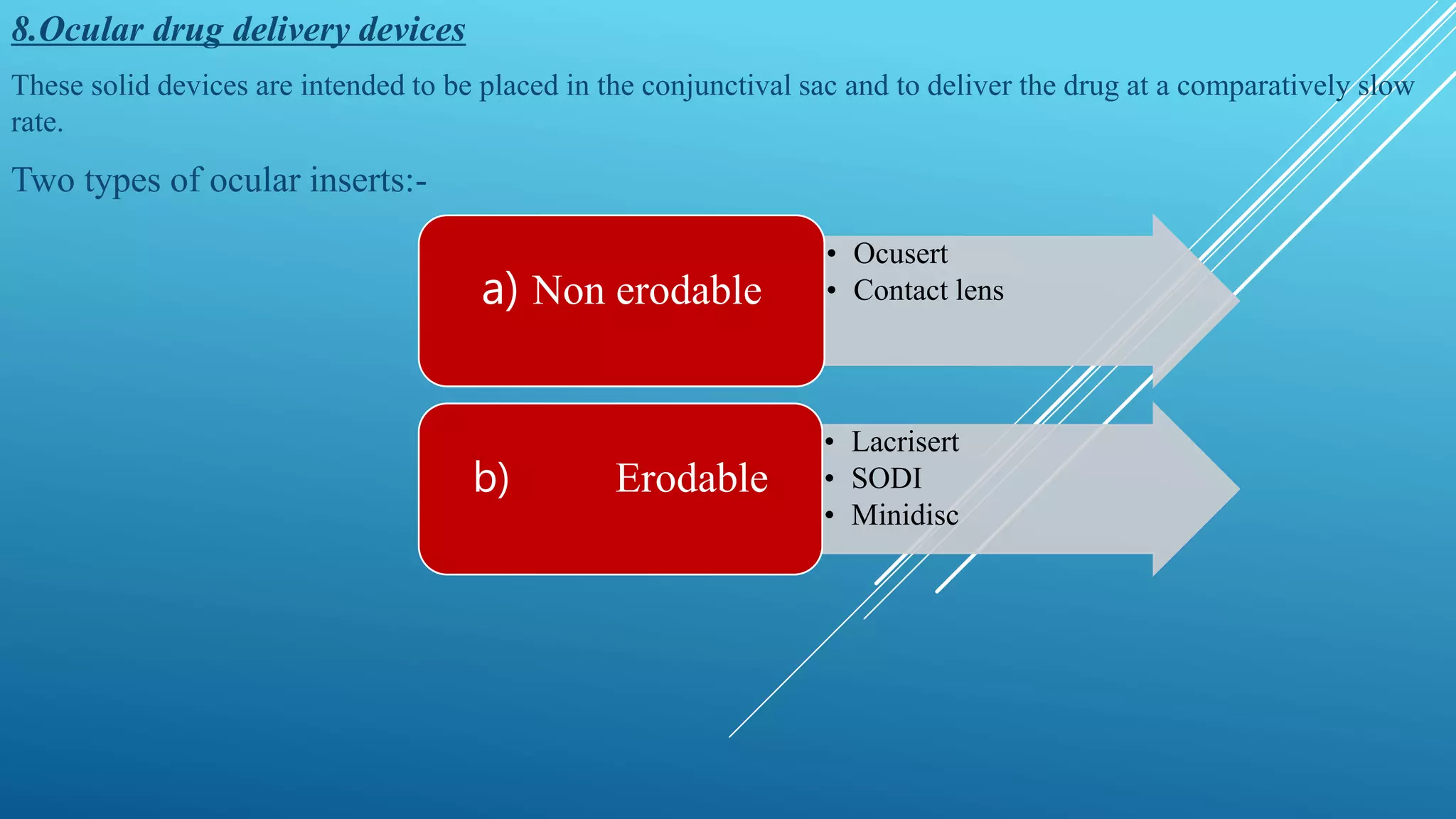
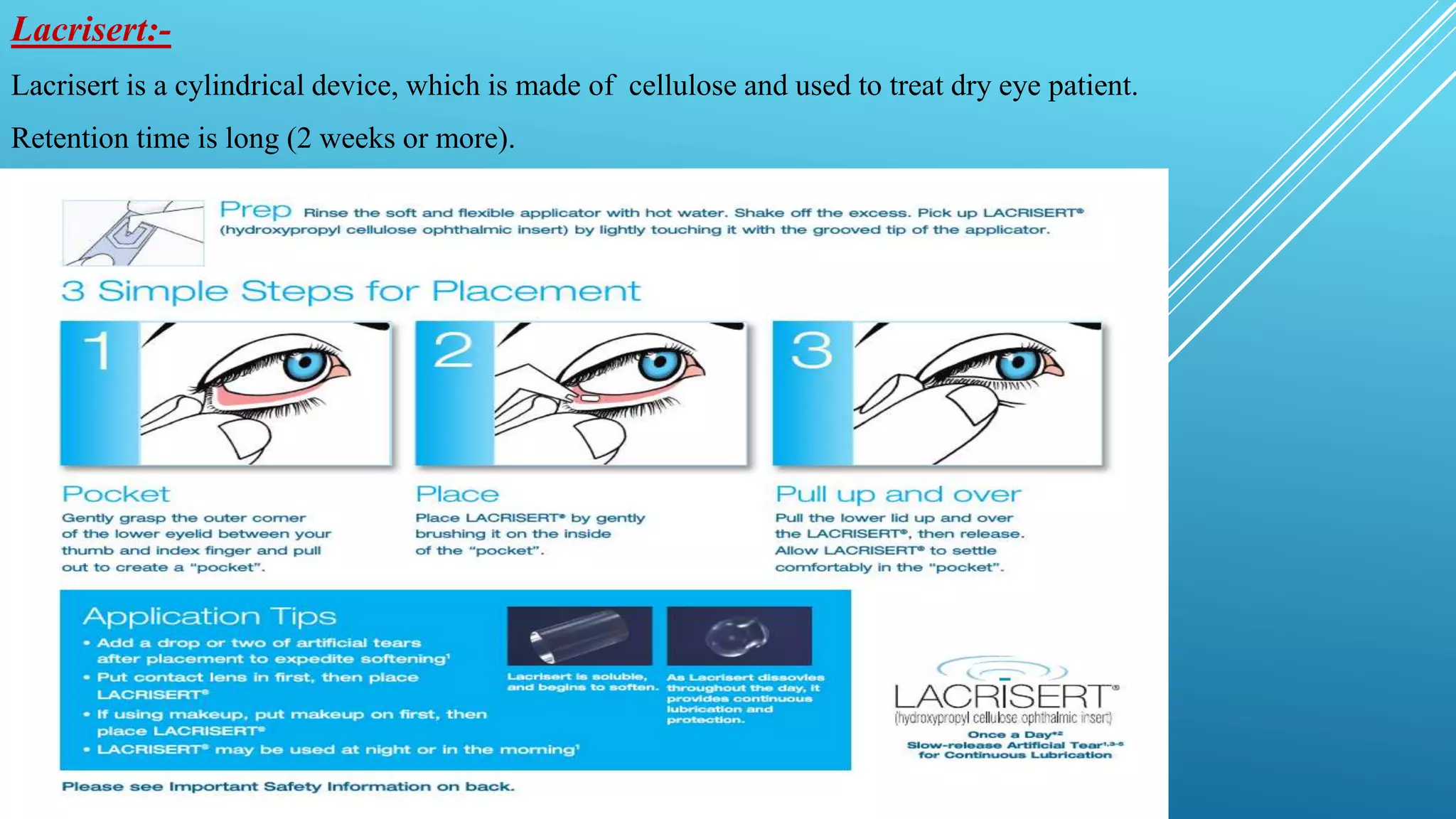
This document discusses ocular drug delivery systems (OCDDS) that aim to prolong drug release in the eye. It introduces various approaches for controlled release, including polymeric solutions, phase transition systems, mucoadhesive dosage forms, collagen shields, and ocular inserts. Specific examples are provided, such as Ocusert which releases pilocarpine at controlled rates over 4-7 days to treat glaucoma. The document outlines the ideal characteristics of OCDDS and mechanisms of controlled drug release via diffusion, osmosis and bioerosion. It also reviews factors influencing ocular drug penetration and absorption.