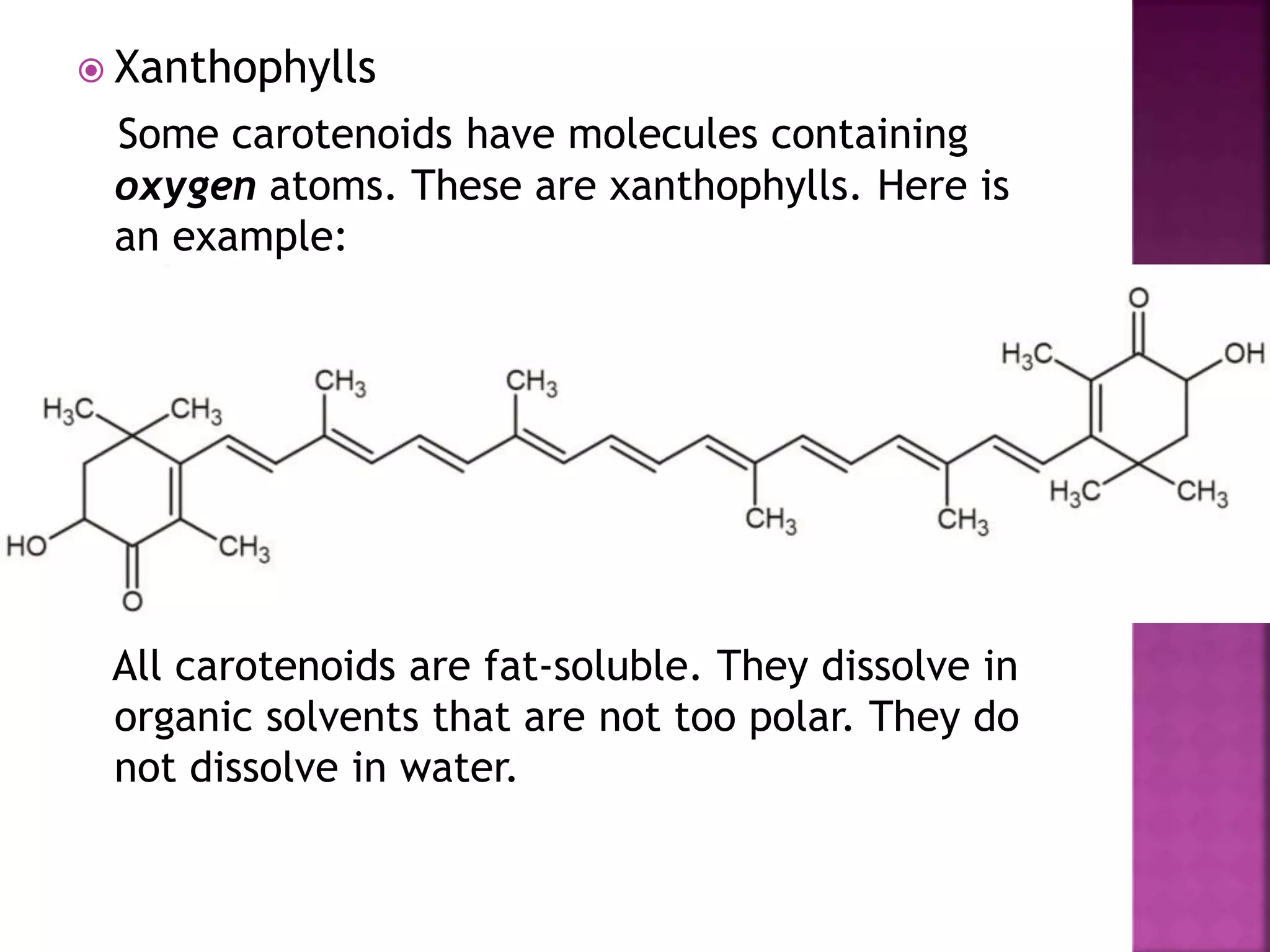
Natural dyes are colorants derived from plants, invertebrates, or minerals. The majority come from plant sources like roots, berries, bark, leaves, and wood. Natural dyes can be grouped according to their chemical structures, such as carotenoids which include carotenes that produce orange and red-orange colors, and xanthophylls which are yellow. Flavonoids are water-soluble compounds that produce yellow, red, violet and blue colors in many flowers and fruits through anthocyanidins and anthocyanins. Anthraquinones and napthoquinones are also groups of natural dyes. Natural dyes have traditionally been used for coloring textiles, leather, and food.